For years, researchers have noted a tantalizing link between some neurologic conditions and certain species of the herpes virus. In patients with Alzheimer’s disease, multiple sclerosis, and cerebellar ataxia, among other neuropathies, the cerebrospinal fluid teems with Epstein-Barr virus (EBV). Yet, the nature of that link has remained unclear, as it has been assumed that EBV, as well as other viruses in the same sub-family, called gammaherpesviruses, cannot infect neurons.
Now, thanks to investigators from the Perelman School of Medicine at the University of Pennsylvania, researchers in this field know better. Erle S. Robertson, PhD, a professor of Microbiology and Otorhinolaryngology and Director of the Tumor Virology Training Program at the Abramson Cancer Center, and colleagues published in mBio this week that EBV and a related virus, Kaposi’s sarcoma-associated herpesvirus (KSHV), can infect and replicate in both cultured and primary neurons.
Though by no means proving causality, those data do suggest viral infection could underlie at least some of the symptoms of those brain disorders, as well as the potential utility of antiviral drugs as a novel therapeutic strategy.
According to Robertson, several lines of evidence suggested the possibility that gammaherpesviruses could infect brain tissue. First, the viruses are enriched in the cerebrospinal fluid and brain tissue of individuals with such conditions as multiple sclerosis (MS) and Alzheimer’s disease. In addition, individuals with a history of infectious mononucleosis caused by EBV are more likely to develop MS, while those who have never been infected with EBV are less likely to do so. Particularly tellingly, the drug acyclovir, which can inhibit EBV and related viruses, has been examined as a potential treatment for MS, with some positive, albeit inconclusive, results.
Still, says Robertson, the ability of gammaherpesviruses to infect neurons has been “controversial.” Devan Mehta, a student in Robertson’s lab, working with postdoctoral fellow Hem C. Jha, PhD, and Dennis Kolson, MD, PhD, a professor of Neurology, tested the link directly. Using genetically modified viruses that express green fluorescent protein (GFP), Mehta infected human neuroblastoma cells (neurons differentiated from cancer cells) and primary human fetal neurons, monitoring the infection over time by microscopy and protein expression.
In both cell types, infection with either EBV or KSHV resulted in the appearance of a fluorescent signal in the infected cells, as well as the appearance of key viral proteins. The media in which infected cells were grown also contained functional virus particles capable of infecting other cells, indicating a mode of infection that tears open host cells. On the other hand, treatment of infected cells with acyclovir reduced the production of virus particles.
“I couldn’t believe it,” Robertson said. “After 50 years of studying EBV, nobody had ever seen the virus in nerve cells. But maybe they just never looked.”
According to Robertson, these data suggest that viral infection of neurons could be associated with neuropathology, though he emphasizes that it is not the same as establishing causality. Such proof, if it ever comes, could be years away.
“There’s likely to be association of this virus with neurons,” he stated. “But more studies will be necessary to know whether it is actually associated with disease pathology.
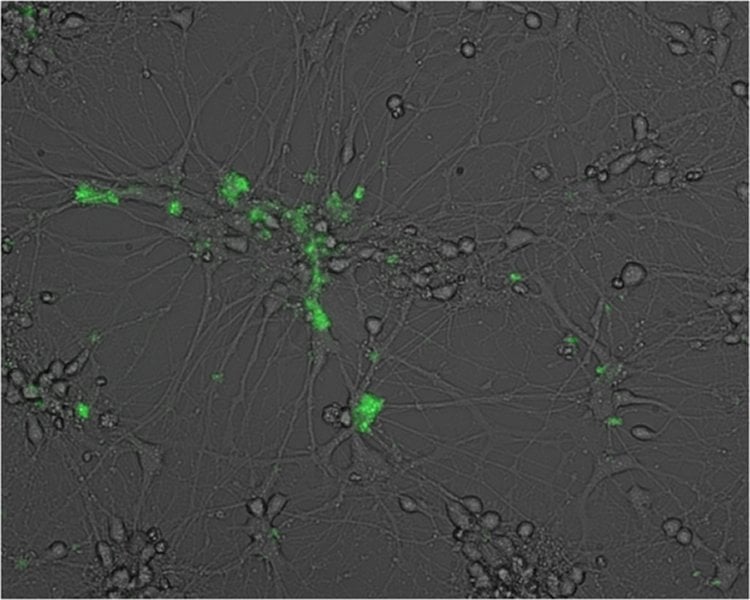
Why EBV and KSHV infection of neurons results in a specific destructive form of infection will also be explored in future research. In contrast, when these viruses infect other cell types, such as B cells, they enter a latent mode, in which virus particles are relatively dormant. But, when they infect neurons, the particles apparently direct the cells to produce large amounts of virus, burst, and die, which explains why the growth media bathing infected cells could be used to infect other cells. “That’s an interesting twist,” Robertson said.
If nothing else, the ability of gammaherpesviruses to infect neurons provides a new model system for studying viral life cycles. But these viruses ultimately may also prove useful in studying disease etiology. “If you can infect nerve cells, that’s likely to have some sort of pathology,” he said.
Additional authors include Jie Lu, Darine El-Naccache, Sanket K. Shukla and Colleen Kovacsics, all from Penn.
Funding: The study was funded by the National Cancer Institute (R01-CA- 137894, R01-CA-171979, R01-CA-177423, CA-137894-05, P30-DK- 050306, and P01-CA-174439), the Leukemia & Lymphoma Society, and the Abramson Cancer Center.
Source: Karen Kreeger – University of Pennsylvania
Image Source: The image is credited to Zhengang Lu
Original Research: Abstract for “Gammaherpesvirus Infection of Human Neuronal Cells” by Hem Chandra Jha, Devan Mehta, Jie Lu, Darine El-Naccache, Sanket K. Shukla, Colleen Kovacsics, Dennis Kolson, and Erle S. Robertson in mBio. Published online December 1 2015 doi:10.1128/mBio.01844-15
Abstract
Gammaherpesvirus Infection of Human Neuronal Cells
Gammaherpesviruses human herpesvirus 4 (HHV4) and HHV8 are two prominent members of the herpesvirus family associated with a number of human cancers. HHV4, also known as Epstein-Barr virus (EBV), a ubiquitous gammaherpesvirus prevalent in 90 to 95% of the human population, is clinically associated with various neurological diseases such as primary central nervous system lymphoma, multiple sclerosis, Alzheimer’s disease, cerebellar ataxia, and encephalitis. However, the possibility that EBV and Kaposi’s sarcoma-associated herpesvirus (KSHV) can directly infect neurons has been largely overlooked. This study has, for the first time, characterized EBV infection in neural cell backgrounds by using the Sh-Sy5y neuroblastoma cell line, teratocarcinoma Ntera2 neurons, and primary human fetal neurons. Furthermore, we also demonstrated KSHV infection of neural Sh-Sy5y cells. These neuronal cells were infected with green fluorescent protein-expressing recombinant EBV or KSHV. Microscopy, genetic analysis, immunofluorescence, and Western blot analyses for specific viral antigens supported and validated the infection of these cells by EBV and KSHV and showed that the infection was efficient and productive. Progeny virus produced from infected neuronal cells efficiently infected fresh neuronal cells, as well as peripheral blood mononuclear cells. Furthermore, acyclovir was effective at inhibiting the production of virus from neuronal cells similar to lymphoblastoid cell lines; this suggests active lytic replication in infected neurons in vitro. These studies represent a potentially new in vitro model of EBV- and KSHV-associated neuronal disease development and pathogenesis.
IMPORTANCE To date, no in vitro study has demonstrated gammaherpesvirus infection of neuronal cells. Moreover, worldwide clinical findings have linked EBV to neuronal pathologies, including multiple sclerosis, primary central nervous system lymphoma, and Alzheimer’s disease. In this study, for the first time, we have successfully demonstrated the in vitro infection of Sh-Sy5y and Ntera2 cells, as well as human primary neurons. We have also determined that the infection is predominately lytic. Additionally, we also report infection of neuronal cells by KSHV in vitro similar to that by EBV. These findings may open new avenues of consideration related to neuronal pathologies and infection with these viruses. Furthermore, their contribution to chronic infection linked to neuronal disease will provide new clues to potential new therapies.
“Gammaherpesvirus Infection of Human Neuronal Cells” by Hem Chandra Jha, Devan Mehta, Jie Lu, Darine El-Naccache, Sanket K. Shukla, Colleen Kovacsics, Dennis Kolson, and Erle S. Robertson in mBio. Published online December 1 2015 doi:10.1128/mBio.01844-15






