Research collaboration uncovers a novel mechanism of altered information processing between neurons. Major relevance for learning and processing of sensory input.
Scientists at Johannes Gutenberg University Mainz (JGU) have discovered a new signal pathway in the brain that plays an important role in learning and the processing of sensory input. It was already known that distinct glial cells receive information from neurons. However, it was unknown that these same glial cells also transmit information to neurons. The glia release a specific protein fragment that influences neuronal cross-talk, most likely by binding to the synaptic contacts that neurons use for communication. Disruption of this information flow from the glia results in changes in the neural network, for example during learning processes. The team composed of Dr. Dominik Sakry, Dr. Angela Neitz, Professor Jacqueline Trotter, and Professor Thomas Mittmann unravelled the underlying mechanism, from the molecular and cellular level to the network and finally the resulting behavioral consequences. Their findings constitute major progress in understanding complex pathways of signal transmission in the brain.
In mammalian brains glial cells outnumber nerve cells, but their functions are still largely unelucidated. A group of glial cells, so-called oligodendrocyte precursor cells (OPC), develop into the oligodendrocytes which ensheathe neuronal axons with a protective myelin layer thus promoting the rapid transmission of signals along the axon. Interestingly, these OPCs are present as a stable proportion – some five to eight percent of all cells in all brain regions, including adult brains. The Mainz-based researchers decided to take a closer look at these OPCs.
In 2000 it was discovered that OPCs receive signals from the neural network via synaptic contacts that they make with neurons. “We have now discovered that the precursor cells do not only receive information via the synapses, but in their turn use these to transmit signals to adjacent nerve cells. They are thus an essential component of the network,” explained Professor Jacqueline Trotter from the Institute of Molecular Cell Biology at Mainz University. Classically, neurons have been considered as the major players in the brain. Over the past few years, however, increasing evidence has come to light that glial cells may play an equally important role. “Glial cells are enormously important for our brains and we have now elucidated in detail a novel important role for glia in signal transmission,” explained Professor Thomas Mittmann of the Institute of Physiology of the Mainz University Medical Center.
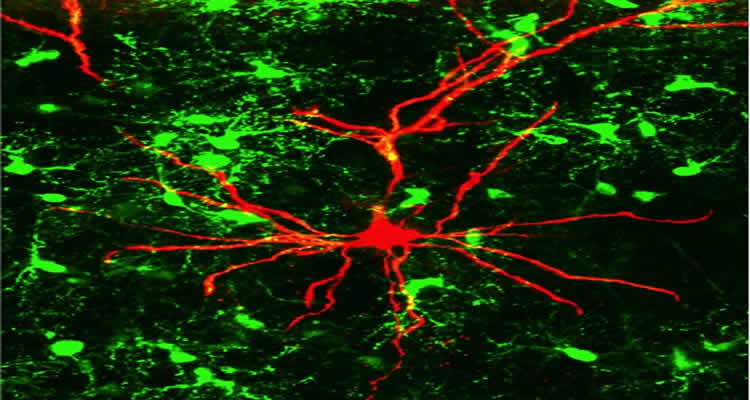
The chain of communication starts with signals traveling from the neurons to the OPCs across the synaptic cleft via the neurotransmitter glutamate. This results in a stimulation of the activity of a specific protease, the alpha-secretase ADAM 10 in OPCs, which acts on the NG2 protein expressed by the precursor cells releasing a NG2 fragment into the extracellular space, where it influences neighboring neuronal synapses. The neurons react to this in the form of altered electrical activity. “We can use patch-clamp techniques to hear, as it were, how the cells talk to one another,” said Mittmann.
“The process starts with the reception of signals coming from the neurons by the OPCs. This means that the feedback to the neurons cannot be seen as separated from the signal reception,” explained Dr. Dominik Sakry, joint first author of the study, describing the cascade of events. The role of NG2 in this process became apparent when the researchers removed the protein: neuronal synaptic function is altered, modifying learning and disrupting the processing of sensory input that manifests in the form of behavioral changes in test animals.

The evidence that the communication between the two cell types in the brain is not a one-way system but a complex mechanism involving feedback loops was obtained in a collaborative project involving physiologists and molecular biologists.
Participating in the project at Mainz University were the Faculties of Biology and Medicine and the Focus Program Translational Neurosciences (FTN) in the form of platform technology provided by the Mouse Behavioral Unit (MBU). The project was additionally supported by two Mainz Collaborative Research Centers (CRC 1080 and CRC-TR 128) and involved participation of the Leibniz Institute for Neurobiology in Magdeburg. Scientists from seven countries participated in the study.
Contact: Dr. Jacqueline Trotter – Johannes Gutenberg Universitaet Mainz
Source: Johannes Gutenberg Universitaet Mainz press release
Image Source: The images are credited to the researchers/Ill.: Institute of Molecular Cell Biology and are adapted from the Johannes Gutenberg Universitaet Mainz press release
Original Research: Full open access research for “Oligodendrocyte Precursor Cells Modulate the Neuronal Network by Activity-Dependent Ectodomain Cleavage of Glial NG2” by Dominik Sakry, Angela Neitz, Jeet Singh, Renato Frischknecht, Daniele Marongiu, Fabien Binamé, Sumudhu S. Perera, Kristina Endres, Beat Lutz, Konstantin Radyushkin, Jacqueline Trotter, and Thomas Mittmann in PLOS Biology. Published online November 11 2014 doi:10.1371/journal.pbio.1001993
Oligodendrocyte Precursor Cells Modulate the Neuronal Network by Activity-Dependent Ectodomain Cleavage of Glial NG2
The role of glia in modulating neuronal network activity is an important question. Oligodendrocyte precursor cells (OPC) characteristically express the transmembrane proteoglycan nerve-glia antigen 2 (NG2) and are unique glial cells receiving synaptic input from neurons. The development of NG2+ OPC into myelinating oligodendrocytes has been well studied, yet the retention of a large population of synapse-bearing OPC in the adult brain poses the question as to additional functional roles of OPC in the neuronal network. Here we report that activity-dependent processing of NG2 by OPC-expressed secretases functionally regulates the neuronal network. NG2 cleavage by the α-secretase ADAM10 yields an ectodomain present in the extracellular matrix and a C-terminal fragment that is subsequently further processed by the γ-secretase to release an intracellular domain. ADAM10-dependent NG2 ectodomain cleavage and release (shedding) in acute brain slices or isolated OPC is increased by distinct activity-increasing stimuli. Lack of NG2 expression in OPC (NG2-knockout mice), or pharmacological inhibition of NG2 ectodomain shedding in wild-type OPC, results in a striking reduction of N-methyl-D-aspartate (NMDA) receptor-dependent long-term potentiation (LTP) in pyramidal neurons of the somatosensory cortex and alterations in the subunit composition of their α-amino-3-hydroxy-5-methyl-4-isoxazolepr opionicacid (AMPA) receptors. In NG2-knockout mice these neurons exhibit diminished AMPA and NMDA receptor-dependent current amplitudes; strikingly AMPA receptor currents can be rescued by application of conserved LNS protein domains of the NG2 ectodomain. Furthermore, NG2-knockout mice exhibit altered behavior in tests measuring sensorimotor function. These results demonstrate for the first time a bidirectional cross-talk between OPC and the surrounding neuronal network and demonstrate a novel physiological role for OPC in regulating information processing at neuronal synapses.
“Oligodendrocyte Precursor Cells Modulate the Neuronal Network by Activity-Dependent Ectodomain Cleavage of Glial NG2” by Dominik Sakry, Angela Neitz, Jeet Singh, Renato Frischknecht, Daniele Marongiu, Fabien Binamé, Sumudhu S. Perera, Kristina Endres, Beat Lutz, Konstantin Radyushkin, Jacqueline Trotter, and Thomas Mittmann in PLOS Biology doi:10.1371/journal.pbio.1001993.






