Summary: Adult neurons in fruitfly brains produce extra copies of their genomes to help prevent cell death caused by DNA damage.
Source: eLife
Scientists have discovered a novel anti-aging defence in the brain cells of adult fruit flies: producing extra copies of the genome, according to a new study published today in eLife.
The findings could help explain how the brain, which rarely produces new cells, is able to cope with the accumulation of cell damage over time and prevent excess cell loss during aging. They may also help scientists answer questions about human age-related brain diseases.
Most cells have two copies of each chromosome, one from each parent. This provides each cell with two complete copies of DNA, referred to as the genome. But some cells accumulate extra copies of chromosomes, although how and why they do this is not entirely clear. Some scientists have suggested that excess chromosomes might lead to age-related brain diseases.
“The brain of the fruit fly Drosophila melanogaster is an ideal model for studying age-related changes in the brain because the fly has a relatively short lifespan, its brain cells rarely multiply, and we have excellent tools for manipulating fly genetics,” says lead author Shyama Nandakumar, a doctoral student in the department of Molecular, Cellular, and Developmental Biology at the University of Michigan, Ann Arbor, US.
In the study, Nandakumar and colleagues examined adult fly brain cells and found that some accumulate extra copies of their genome, especially in parts of the brain responsible for vision – the region of the brain which shows more DNA damage with age.
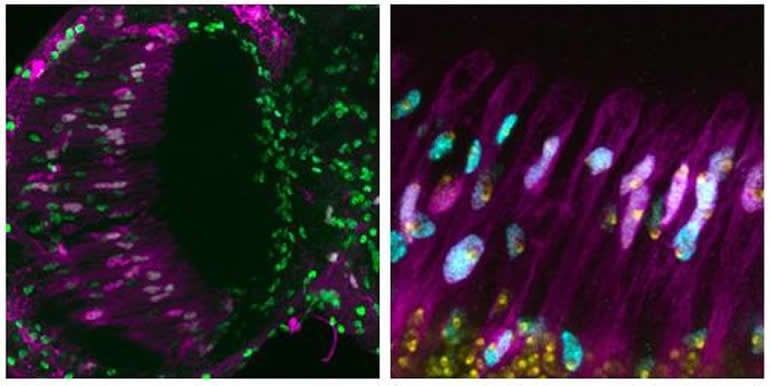
Next, they subjected fly brain cells to oxidative stress or ultraviolet radiation, which damage DNA and can cause cell death. They found that this exposure increased the production of extra copies of chromosomes in the cells, and the cells were less likely to die as a result of the damage. “These data suggest that cells with extra copies of the genome are more resistant to cell death and may serve a beneficial or protective role in the aging brain,” Nandakumar explains.
Previous studies have found that patients with early stages of Alzheimer’s disease have extra chromosomes in their brain cells than people of the same age who do not have the condition. This has led scientists to question whether accumulating extra chromosomes leads to brain cell death and brain disease.
“Our study suggests that the production of extra copies of chromosomes might actually be a normal response to the accumulation of age-related damage in flies and may even help protect against cell death,” concludes senior author Laura Buttitta, Associate Professor of Molecular, Cellular and Developmental Biology at the University of Michigan. “Further studies are now needed to determine if this is also true in humans.”
About this genetics research article
Source:
eLife
Contacts:
Emily Packer – eLife
Image Source:
The image is credited to Nandakumar et al.
Original Research: Open access
“Polyploidy in the adult Drosophila brain” by Shyama Nandakumar, Olga Grushko, Laura A Buttitta. eLife.
Abstract
Polyploidy in the adult Drosophila brain
Long-lived cells such as terminally differentiated postmitotic neurons and glia must cope with the accumulation of damage over the course of an animal’s lifespan. How long-lived cells deal with ageing-related damage is poorly understood. Here we show that polyploid cells accumulate in the adult fly brain and that polyploidy protects against DNA damage-induced cell death. Multiple types of neurons and glia that are diploid at eclosion, become polyploid in the adult Drosophila brain. The optic lobes exhibit the highest levels of polyploidy, associated with an elevated DNA damage response in this brain region. Inducing oxidative stress or exogenous DNA damage leads to an earlier onset of polyploidy, and polyploid cells in the adult brain are more resistant to DNA damage-induced cell death than diploid cells. Our results suggest polyploidy may serve a protective role for neurons and glia in adult Drosophila melanogaster brains.






