Abuse of prescription opioid medicines used to treat chronic pain has reached epidemic proportions, so much that the White House has announced new efforts to combat addiction and prevent the thousands of overdose-related deaths reported in the U.S. each year.
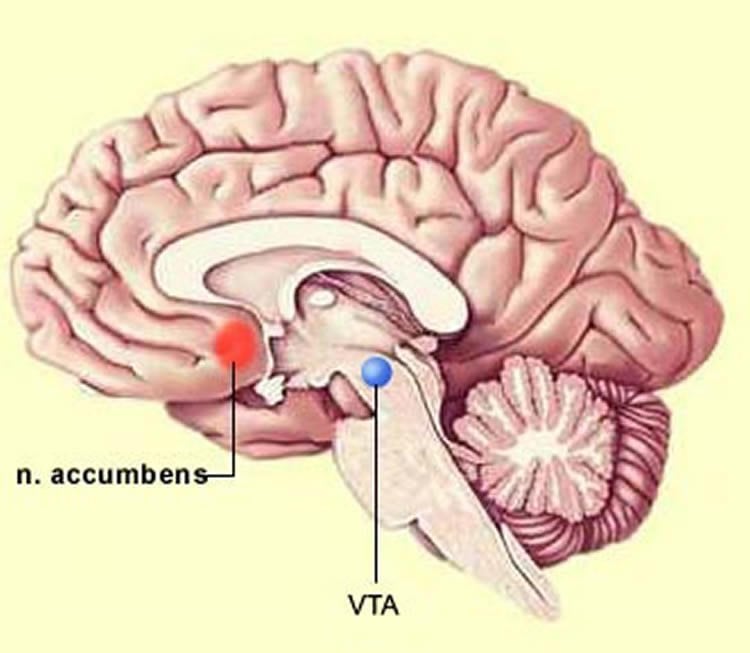
But a University of Texas at Arlington research team has been working on an alternative solution: electrical stimulation of a deep, middle brain structure that blocks pain signals at the spinal cord level without drug intervention. The process also triggers the release of beneficial dopamine, which may reduce the emotional distress associated with long-term pain, researchers said.
“This is the first study to use a wireless electrical device to alleviate pain by directly stimulating the ventral tegmental area of the brain,” said Yuan Bo Peng, UTA psychology professor. “While still under laboratory testing, this new method does provide hope that in the future we will be able to alleviate chronic pain without the side effects of medications.”
Peng and J.-C. Chiao, an electrical engineering professor, detail their discoveries in a new paper published in the leading neuroscience journal Experimental Brain Research. Professor Xiaofei Yang, an electrical engineering professor at Huazhong University of Science and Technology in Wuhan, China also participated in the study.
The project was supported partly by grants received from the Texas Norman Hackerman Advanced Research Program, Intel Corp. and the Texas Medical Research Collaborative, a research partnership among universities, health care providers and corporations that gives grants to jump-start research around real-world problems.
Nearly two million Americans abused or were dependent on opioid medicines in 2014, and 165,000 died between 1999 and 2014 from overdoses related to opioid prescriptions, according to the Centers for Disease Control.
In their experiments, Peng and Chiao used their patented custom-designed wireless device to demonstrate that stimulation of the ventral tegmental area reduced the sensation of pain. They also confirmed that this stimulation reduced pain signals in the spinal cord, effectively blocking the perception of pain.
Morteza Khaledi, dean of the UTA’s College of Science, commended the researchers on this important work.
“Solutions for chronic pain are at the forefront of current medical research,” Khaledi said. “Dr. Peng and Dr. Chiao’s research is high-impact work focused on health and the human condition, a key theme within UTA’s Strategic Plan 2020: Bold Solutions | Global Impact.”
Chiao has constant pain from slipped discs and pinched nerves in his neck and also witnessed his uncle suffer after chemotherapy for prostate cancer. His uncle used an early implant device to electrically stimulate his spinal cord and relieve the pain. The technology was the best available, but he had to manually change the stimulation dosages every 15 minutes, and suffered without much sleep before he passed away.
These difficult experiences have fueled Chiao and Peng’s decade-long commitment to finding a solution for chronic pain by devising advanced wireless implants that take patients out of the equation, treating their pain in the background as they go on with their lives. Dr. Peng initiated the idea and worked with Chiao, Yang and other researchers to develop new technology, methods and knowledge across disciplines.
Chiao earned his doctorate in electrical engineering from California Institute of Technology and holds several patents, including in the area of wireless medical sensor technologies. Currently, he is the Janet and Mike Greene Endowed Professor and Jenkins Garrett endowed Professor of Electrical Engineering and Joint Biomedical Engineering Program at UTA.
Peng has specialized in pain relief throughout his career. He was a medical doctor and holds a doctorate in neuroscience from University of Texas Medical Branch in Galveston. He has held postdoctoral fellowships focused on pain at Johns Hopkins Medical Institute, as well as the National Institute of Dental and Craniofacial Research, the National Institutes of Health, and the Department of Health and Human Services.
Other participants in this research were Ai-Ling Li, a postdoctoral fellow in Indiana University who earned her psychology doctorate with Peng at UTA, and Jiny Sibi, a medical student at the University of Texas Medical Branch in Galveston, who was previously an undergraduate student in Peng’s laboratory at UTA.
“Until this study, the ventral segmental area of the brain was studied more for its key role in positive reinforcement, reward and drug abuse,” said Peng. “We have now confirmed that stimulation of this area of the brain can also be an analgesic tool.”
Source: Louisa Kellie – UT Arlington
Image Source: The image is credited to McGill University and is licensed CC BY SA 3.0.
Original Research: Abstract for “Stimulation of the ventral tegmental area increased nociceptive thresholds and decreased spinal dorsal horn neuronal activity in rat” by Ai-Ling Li, Jiny E. Sibi, Xiaofei Yang, Jung-Chih Chiao, and Yuan Bo Peng in Experimental Brain Research. Published online January 28 2016 doi:10.1007/s00221-016-4558-z
Abstract
Stimulation of the ventral tegmental area increased nociceptive thresholds and decreased spinal dorsal horn neuronal activity in rat
Deep brain stimulation has been found to be effective in relieving intractable pain. The ventral tegmental area (VTA) plays a role not only in the reward process, but also in the modulation of nociception. Lesions of VTA result in increased pain thresholds and exacerbate pain in several pain models. It is hypothesized that direct activation of VTA will reduce pain experience. In this study, we investigated the effect of direct electrical stimulation of the VTA on mechanical, thermal and carrageenan-induced chemical nociceptive thresholds in Sprague–Dawley rats using our custom-designed wireless stimulator. We found that: (1) VTA stimulation itself did not show any change in mechanical or thermal threshold; and (2) the decreased mechanical and thermal thresholds induced by carrageenan injection in the hind paw contralateral to the stimulation site were significantly reversed by VTA stimulation. To further explore the underlying mechanism of VTA stimulation-induced analgesia, spinal cord dorsal horn neuronal responses to graded mechanical stimuli were recorded. VTA stimulation significantly inhibited dorsal horn neuronal activity in response to pressure and pinch from the paw, but not brush. This indicated that VTA stimulation may have exerted its analgesic effect via descending modulatory pain pathways, possibly through its connections with brain stem structures and cerebral cortex areas.
“Stimulation of the ventral tegmental area increased nociceptive thresholds and decreased spinal dorsal horn neuronal activity in rat” by Ai-Ling Li, Jiny E. Sibi, Xiaofei Yang, Jung-Chih Chiao, and Yuan Bo Peng in Experimental Brain Research. Published online January 28 2016 doi:10.1007/s00221-016-4558-z







