Summary: CRISPR gene editing has allowed researchers to prevent angiogenesis of the retina occurring in mice. Researchers say the new technology could help prevent vision loss in those with retinal diseases.
Source: Mass Eye and Ear.
Powerful new technology may lead to novel therapies to prevent vision loss, blindness in those with diseases of the retina.
A research team from the Schepens Eye Research Institute of Massachusetts Eye and Ear has successfully prevented mice from developing angiogenesis of the retina—the sensory tissue at the back of the eye—using gene-editing techniques with CRISPR-Cas9. Angiogenesis causes vision loss and blindness and is a feature of several degenerative eye conditions, including proliferative diabetic retinopathy (PDR), wet age-related macular degeneration (AMD), and retinopathy of prematurity (ROP). In a report published online today in Nature Communications, the researchers present a novel gene-editing technique to prevent retinal angiogenesis, which could lead to the development of new therapies for eye conditions marked by pathological intraocular angiogenesis.
Despite the success of vascular endothelial cell growth factor (VEGF) inhibiting agents (e.g. Lucentis®, Eylea®) in reducing neovascular growth and lessening vascular leakage in retinal diseases such as PDR and AMD, several therapeutic challenges remain—namely a need for sustained treatment and a modality to treat the significant number of patients who do not respond to anti-VEGF therapies.
“We know that vascular endothelial growth factor (VEGF) receptor 2 (VEGFR2) plays an essential role in angiogenesis,” said corresponding author Hetian Lei, Ph.D., Assistant Scientist at Schepens Eye Research Institute of Mass. Eye and Ear and Assistant Professor of Ophthalmology at Harvard Medical School. “The CRISPR-Cas9 system to can be utilized to edit the VEGFR2 gene, preventing intraocular pathological angiogenesis.”
A feature of various eye diseases, pathological intraocular angiogenesis presents clinically when blood vessels in the retina (the structure in the back of the eye that senses and perceives light) begin to grow new, abnormal blood vessels on the surface of the retina. As the damage progresses, these vessels can leak, rupture, or cause retinal detachment leading to impaired vision.
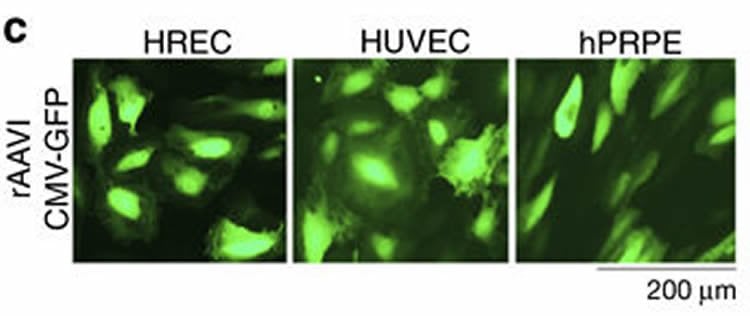
CRISPR-Cas9 is a powerful new technology that can target and edit certain aspects of the genome, or the complete set of genetic material of an organism. In the Nature Communications report, study authors used an adeno-associated virus (AAV) to deliver genomic edits to target VEGFR2, a critical protein responsible for angiogenesis. A single injection of this therapy was able to prevent retinal angiogenesis in preclinical models.
“As this genomic editing gains traction in virtually all medical fields, we are cautiously optimistic that this powerful tool may present a novel therapy to prevent vision loss in eye disease marked by intraocular pathological angiogenesis,” said Dr. Lei. “While further study is needed to determine safety and efficacy of this approach, our work shows that the CRISPR-Cas9 system is a precise and efficient tool with the potential to treat angiogenesis-associated diseases.”
In addition to Dr. Lei, authors on the Nature Communications paper include Xionggao Huang, Ph.D., Guohong Zhou, Ph.D., Wenyi Wu, Yajian Duan, Gaoen Ma, Jingyuan Song, Ph.D., Ru Xiao, Ph.D., Luk Vandenberghe, Ph.D., and Patricia D’Amore, Ph.D., of the Schepens Eye Research Institute of Mass. Eye and Ear, as well as Feng Zhang, Ph.D., of the Broad Institute of the Massachusetts Institute of Technology and Harvard University.
Funding: This research study was supported by National Institutes of Health/National Eye Institute grants R01EY012509 and P30EY003790.
Source: Suzanne Day – Mass Eye and Ear
Image Source: NeuroscienceNews.com image is credited to Lei et al./Nature Communications.
Original Research: Full open access research for “Genome editing abrogates angiogenesis in vivo” by Xionggao Huang, Guohong Zhou, Wenyi Wu, Yajian Duan, Gaoen Ma, Jingyuan Song, Ru Xiao, Luk Vandenberghe, Feng Zhang, Patricia A. D’Amore & Hetian Lei in Nature Communications. Published online July 24 2017 doi:10.1038/s41467-017-00140-3
[cbtabs][cbtab title=”MLA”]Mass Eye and Ear “Genome Editing with CRISPR-Cas9 Prevents Angiogenesis of the Retina.” NeuroscienceNews. NeuroscienceNews, 26 July 2017.
<https://neurosciencenews.com/crispr-angiogenesis-retina-7184/>.[/cbtab][cbtab title=”APA”]Mass Eye and Ear (2017, July 26). Genome Editing with CRISPR-Cas9 Prevents Angiogenesis of the Retina. NeuroscienceNew. Retrieved July 26, 2017 from https://neurosciencenews.com/crispr-angiogenesis-retina-7184/[/cbtab][cbtab title=”Chicago”]Mass Eye and Ear “Genome Editing with CRISPR-Cas9 Prevents Angiogenesis of the Retina.” https://neurosciencenews.com/crispr-angiogenesis-retina-7184/ (accessed July 26, 2017).[/cbtab][/cbtabs]
Abstract
Genome editing abrogates angiogenesis in vivo
Angiogenesis, in which vascular endothelial growth factor receptor (VEGFR) 2 plays an essential role, is associated with a variety of human diseases including proliferative diabetic retinopathy and wet age-related macular degeneration. Here we report that a system of adeno-associated virus (AAV)-mediated clustered regularly interspaced short palindromic repeats (CRISPR)-associated endonuclease (Cas)9 from Streptococcus pyogenes (SpCas9) is used to deplete VEGFR2 in vascular endothelial cells (ECs), whereby the expression of SpCas9 is driven by an endothelial-specific promoter of intercellular adhesion molecule 2. We further show that recombinant AAV serotype 1 (rAAV1) transduces ECs of pathologic vessels, and that editing of genomic VEGFR2 locus using rAAV1-mediated CRISPR/Cas9 abrogates angiogenesis in the mouse models of oxygen-induced retinopathy and laser-induced choroid neovascularization. This work establishes a strong foundation for genome editing as a strategy to treat angiogenesis-associated diseases.
“Genome editing abrogates angiogenesis in vivo” by Xionggao Huang, Guohong Zhou, Wenyi Wu, Yajian Duan, Gaoen Ma, Jingyuan Song, Ru Xiao, Luk Vandenberghe, Feng Zhang, Patricia A. D’Amore & Hetian Lei in Nature Communications. Published online July 24 2017 doi:10.1038/s41467-017-00140-3






