Summary: Researchers use PET neuroimaging technology to help determin which children with DIPG brain cancer will benefit best from Avastin.
Source: Society of Nuclear Medicine.
Brain cancers are difficult to treat, and it can be hard to predict whether a therapy will be effective. When the patient is a child, it’s even more important to predict the potential effectiveness of a drug before beginning treatment. In this first ever molecular drug-imaging study in children, researchers in The Netherlands used whole-body positron emission tomography/computed tomography (PET/CT) scans to determine whether bevacizumab (Avastin) treatment of diffuse intrinsic pontine glioma (DIPG) in children is likely to be effective. The study is featured in the May 2017 issue of The Journal of Nuclear Medicine.
“Children with DIPG have a very poor prognosis, with less than 10 percent of the patients surviving two years from diagnosis,” explains Guus A. van Dongen, PhD, of VU University, Medical Center, Amsterdam, The Netherlands. “These tumors are resistant to all kinds of therapies. Chemotherapy, as well as new targeted therapies, may not reach the tumor due to the location within the brainstem.”
For the study, researchers investigated whether bevacizumab can reach the tumor in children with DIPG by measuring the tumor uptake of zirconium-89 (Zr-89)-labeled bevacizumab with PET. In addition, they evaluated the safety of the procedure and determined the optimal timing of imaging.
Two weeks after completing radiotherapy, seven patients (age range: 6-17) were given whole-body PET/CT scans performed at 1, 72 and 144 hours post-injection. The optimal moment of scanning was found to be 144 hours post-injection. The patients also underwent contrast (gadolinium)-enhanced MRI.
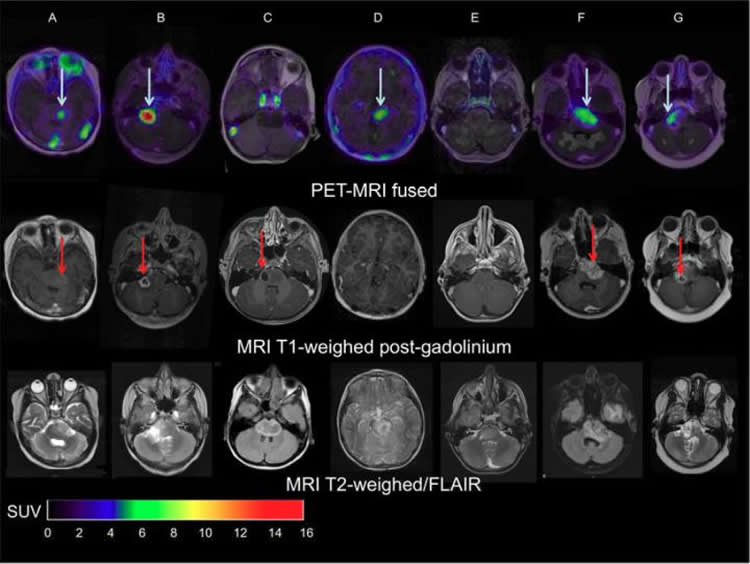
“The results showed that indeed there is considerable heterogeneity in uptake of Zr-89-labeled bevacizumab among patients and within tumors,” Van Dongen points out. “This non-invasive in vivo quantification of drug distribution and tumor uptake may help to predict therapeutic potential, as well as toxicity, and could help develop strategies for improving drug delivery to tumors.”
Van Dongen adds, “Children with brain tumors and other solid cancers are particularly likely to benefit from molecular drug imaging, as drugs without therapeutic effect–based on a lack of drug-uptake in the tumor–may cause life-long side effects. Molecular drug imaging will open avenues for administering the right drug to the right patient at the most appropriate stage of the disease.”
Funding: Funding for this study was provided by the Semmy Foundation and Egbers Foundation.
Source: Laurie Callahan – Society of Nuclear Medicine
Image Source: NeuroscienceNews.com image is credited to Sophie Veldhuijzen van Zanten and Marc Jansen, VU University Medical Center, Amsterdam, The Netherlands.
Original Research: Abstract for “Molecular Drug Imaging: 89Zr-Bevacizumab PET in Children with Diffuse Intrinsic Pontine Glioma” by Marc H. Jansen, Sophie E.M. Veldhuijzen van Zanten, Dannis G. van Vuurden, Marc C. Huisman, Danielle J. Vugts, Otto S. Hoekstra, Guus A. van Dongen, and Gert-Jan L. Kaspers in Journal of Nuclear Medicine. Published online May 1 2017 doi:10.2967/jnumed.116.180216
[cbtabs][cbtab title=”MLA”]Society of Nuclear Medicine “PET/CT Helps Predict Therapy Effectiveness in Pediatric Brain Tumors.” NeuroscienceNews. NeuroscienceNews, 2 May 2017.
<https://neurosciencenews.com/brain-scan-pediatric-cancer-6568/>.[/cbtab][cbtab title=”APA”]Society of Nuclear Medicine (2017, May 2). PET/CT Helps Predict Therapy Effectiveness in Pediatric Brain Tumors. NeuroscienceNew. Retrieved May 2, 2017 from https://neurosciencenews.com/brain-scan-pediatric-cancer-6568/[/cbtab][cbtab title=”Chicago”]Society of Nuclear Medicine “PET/CT Helps Predict Therapy Effectiveness in Pediatric Brain Tumors.” https://neurosciencenews.com/brain-scan-pediatric-cancer-6568/ (accessed May 2, 2017).[/cbtab][/cbtabs]
Abstract
Molecular Drug Imaging: 89Zr-Bevacizumab PET in Children with Diffuse Intrinsic Pontine Glioma
Predictive tools for guiding therapy in children with brain tumors are urgently needed. In this first molecular drug imaging study in children, we investigated whether bevacizumab can reach tumors in children with diffuse intrinsic pontine glioma (DIPG) by measuring the tumor uptake of 89Zr-labeled bevacizumab by PET. In addition, we evaluated the safety of the procedure in children and determined the optimal time for imaging.
Methods: Patients received 89Zr-bevacizumab (0.1 mg/kg; 0.9 MBq/kg) at least 2 wk after completing radiotherapy. Whole-body PET/CT scans were obtained 1, 72, and 144 h after injection. All patients underwent contrast (gadolinium)-enhanced MRI. The biodistribution of 89Zr-bevacizumab was quantified as SUVs.
Results: Seven DIPG patients (4 boys; 6–17 y old) were scanned without anesthesia. No adverse events occurred. Five of 7 primary tumors showed focal 89Zr-bevacizumab uptake (SUVs at 144 h after injection were 1.0–6.7), whereas no significant uptake was seen in the healthy brain. In 1 patient, multiple metastases all showed positive PET results. We observed inter- and intratumoral heterogeneity of uptake, and 89Zr-bevacizumab uptake was present predominantly (in 4/5 patients) within MRI contrast-enhanced areas, although 89Zr-bevacizumab uptake in these areas was variable. Tumor targeting results were quantitatively similar at 72 and 144 h after injection, but tumor–to–blood-pool SUV ratios increased with time after injection (P = 0.045). The mean effective dose per patient was 0.9 mSv/MBq (SD, 0.3 mSv/MBq).
Conclusion: 89Zr-bevacizumab PET studies are feasible in children with DIPG. The data suggest considerable heterogeneity in drug delivery among patients and within DIPG tumors and a positive, but not 1:1, correlation between MRI contrast enhancement and 89Zr-bevacizumab uptake. The optimal time for scanning is 144 h after injection. Tumor 89Zr-bevacizumab accumulation assessed by PET scanning may help in the selection of patients with the greatest chance of benefit from bevacizumab treatment.
“Molecular Drug Imaging: 89Zr-Bevacizumab PET in Children with Diffuse Intrinsic Pontine Glioma” by Marc H. Jansen, Sophie E.M. Veldhuijzen van Zanten, Dannis G. van Vuurden, Marc C. Huisman, Danielle J. Vugts, Otto S. Hoekstra, Guus A. van Dongen, and Gert-Jan L. Kaspers in Journal of Nuclear Medicine. Published online May 1 2017 doi:10.2967/jnumed.116.180216






