Summary: Researchers have identified a specific pathway between the hypothalamus and habenula that controls feelings of aversion.
Source: Karolinska Institute.
What happens in the brain when we feel discomfort? Researchers at Karolinska Institutet in Sweden are now one step closer to finding the answer. In a new study published in the journal Molecular Psychiatry they identify which pathways in the mouse brain control behaviour associated with aversion.
Scientists have long been interested in how the brain creates signals associated with negative emotions in order to better understand how imbalances in the same system can lead to affective disorders such as depression and anxiety.
The amygdala has long been the most commonly studied brain structure for understanding fear, whereas for rewards and positive signals the focus has been on the neurotransmitter dopamine. But when it comes to areas of the brain that control feelings of discomfort and aversion, much less is known.
In the past few years, research has indicated that a brain structure called the habenula controls positive and negative emotions in animal models. Moreover, clinical cases have been conducted with patients suffering from depression where deep brain stimulation of the habenula has been beneficial. The habenula controls both dopamine and the neurotransmitter serotonin, which is thought to play a significant part in the sense of wellbeing. However, it has not been known how the habenula is regulated.
Researchers at Karolinska Institutet have now mapped which networks in the mouse brain control the habenula, and what role they play in aversion.
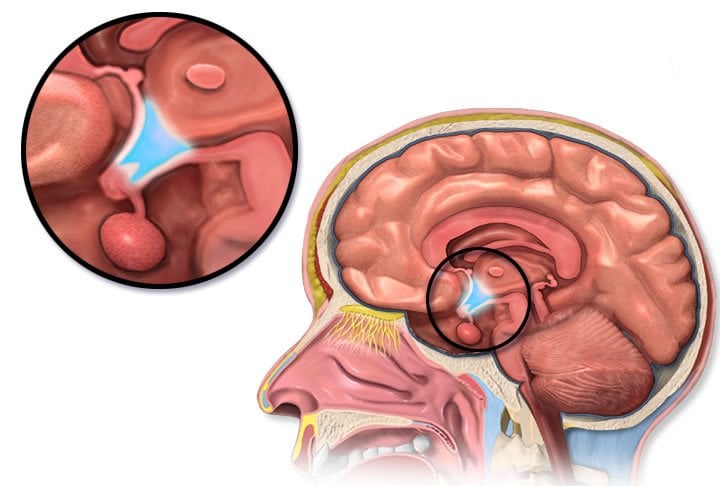
“We’ve discovered a specific pathway that goes between the hypothalamus and the habenula, and that can be modulated using optogenetics to control the feeling of aversion,” says study leader docent Dinos Meletis at the Department of Neuroscience. “Our hope is that this can lead to the development of new treatments that can rebalance the brain’s networks in for example depression or anxiety disorders.”
Using optogenetics and other advanced methods, the group were able to identify the identity of the nerve cells and map their interconnections. Optogenetics is a method that uses light to activate specific neurons in order to study how the activation of different networks affects behaviour.
“This methodological revolution in brain research has made it possible to functionally study how different types of nerve cell and pathways actually control different types of behaviour, something that was impossible to do only a decade ago,” says Dr Meletis.
Funding: The study was supported by grants from the Swedish Research Council, the Swedish Foundation for Strategic Research, the Swedish Brain Foundation and Karolinska Institutet.
Source: Karolinska Institute
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to BruceBlaus. Licensed CC by 3.0.
Original Research: Open access research for “A hypothalamus-habenula circuit controls aversion” by Iakovos Lazaridis, Ourania Tzortzi, Moritz Weglage, Antje Märtin, Yang Xuan, Marc Parent, Yvonne Johansson, Janos Fuzik, Daniel Fürth, Lief E. Fenno, Charu Ramakrishnan, Gilad Silberberg, Karl Deisseroth, Marie Carlén & Konstantinos Meletis in Molecular Psychiatry. Published February 12 2019.
doi:10.1159/000496086
[cbtabs][cbtab title=”MLA”]Karolinska Institute”Brain Pathways of Aversion Identified.” NeuroscienceNews. NeuroscienceNews, 14 February 2019.
<https://neurosciencenews.com/aversion-pathway-10739/>.[/cbtab][cbtab title=”APA”]Karolinska Institute(2019, February 14). Brain Pathways of Aversion Identified. NeuroscienceNews. Retrieved February 14, 2019 from https://neurosciencenews.com/aversion-pathway-10739/[/cbtab][cbtab title=”Chicago”]Karolinska Institute”Brain Pathways of Aversion Identified.” https://neurosciencenews.com/aversion-pathway-10739/ (accessed February 14, 2019).[/cbtab][/cbtabs]
Abstract
A hypothalamus-habenula circuit controls aversion
Encoding and predicting aversive events are critical functions of circuits that support survival and emotional well-being. Maladaptive circuit changes in emotional valence processing can underlie the pathophysiology of affective disorders. The lateral habenula (LHb) has been linked to aversion and mood regulation through modulation of the dopamine and serotonin systems. We have defined the identity and function of glutamatergic (Vglut2) control of the LHb, comparing the role of inputs originating in the globus pallidus internal segment (GPi), and lateral hypothalamic area (LHA), respectively. We found that LHb-projecting LHA neurons, and not the proposed GABA/glutamate co-releasing GPi neurons, are responsible for encoding negative value. Monosynaptic rabies tracing of the presynaptic organization revealed a predominantly limbic input onto LHA Vglut2 neurons, while sensorimotor inputs were more prominent onto GABA/glutamate co-releasing GPi neurons. We further recorded the activity of LHA Vglut2 neurons, by imaging calcium dynamics in response to appetitive versus aversive events in conditioning paradigms. LHA Vglut2 neurons formed activity clusters representing distinct reward or aversion signals, including a population that responded to mild foot shocks and predicted aversive events. We found that the LHb-projecting LHA Vglut2 neurons encode negative valence and rapidly develop a prediction signal for negative events. These findings establish the glutamatergic LHA-LHb circuit as a critical node in value processing.