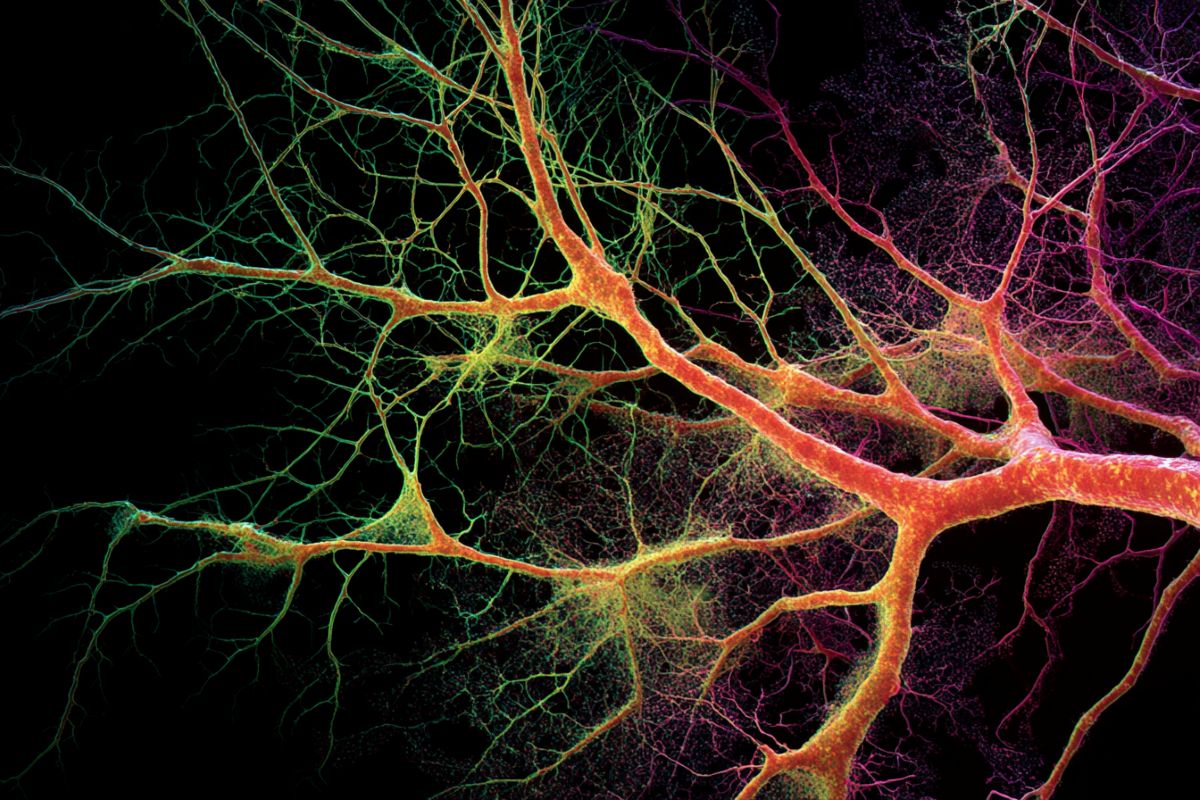
Summary: When we learn a new motor skill, we usually think of neurons doing all the work. However, a groundbreaking study reveals that astrocytes—star-shaped non-neuronal cells—are actually the master “pruners” of the brain’s motor circuits.
The team discovered that astrocytes in the striatum use a receptor called MEGF10 to physically eliminate unnecessary synapses. This pruning process is triggered by dopamine and neural activity, proving that astrocytes aren’t just support cells; they are essential for translating dopamine signals into the lasting structural changes required to master a new skill.
Key Facts
- Active Pruning: Astrocytes actively remove synapses in the striatum during motor learning to refine neural circuits.
- The MEGF10 Receptor: This specific receptor on astrocytes acts as the “mouth” that engulfs and eliminates unnecessary synaptic connections.
- Dopamine Regulation: Dopamine doesn’t just signal reward; it tells astrocytes which synapses to remove and which to save, specifically affecting D1 and D2 medium spiny neurons.
- Learning Impairment: Mice lacking the MEGF10 receptor in their astrocytes were significantly worse at learning motor tasks (like the rotarod test) and showed disrupted communication between the motor cortex and striatum.
- Plasticity Hub: The study shows that both long-term potentiation (LTP) and long-term depression (LTD) require astrocytic participation to function correctly during learning.
Source: Institute for Basic Science
When we learn a new motor skill—whether mastering a piano passage or refining balance while walking—the brain must reorganize the circuits that control movement. For decades, this process of synaptic remodeling has been attributed primarily to neurons strengthening or weakening their connections.
However, the new study reveals that another cell type in the brain called astrocytes actively participates in this rewiring process.
A research team led by CHUNG Won-Suk (KAIST Department of Biological Sciences), Associate Director of the Center for Vascular Research within the Institute for Basic Science (IBS), and Professor KIM Jae-Ick at UNIST has demonstrated that astrocytes actively eliminate synapses in the striatum, a brain region that plays a central role in controlling voluntary movement and learning.
This process is regulated by dopamine signaling and neural activity and is critical for proper motor skill acquisition.
Although synapse formation and elimination have long been studied in the context of neuronal plasticity, increasing evidence suggests that glial cells—particularly astrocytes and microglia—also contribute to synapse turnover.
Until now, however, the precise role of astrocytes in motor learning and the mechanisms underlying their synaptic remodeling remained unclear.
To address this question, the researchers used mouse models undergoing repeated motor training tasks, including the rotarod test, which measures motor coordination and learning.
Using advanced imaging tools that can track individual synaptic components, the team observed a marked increase in astrocyte-mediated synapse elimination as motor learning progressed.
In contrast, other glial cell types, such as microglia and oligodendrocyte precursor cells, showed no significant changes under the same experimental conditions, indicating a specific role for astrocytes in this process.
The researchers identified MEGF10, a phagocytic receptor expressed in astrocytes, as a key molecular mediator of this remodeling. When MEGF10 was selectively deleted in astrocytes, mice exhibited impaired motor learning and significant disruptions in communication between the motor cortex and the striatum.
In addition, both long-term potentiation (LTP) and long-term depression (LTD)—two fundamental mechanisms of synaptic plasticity—were compromised. These results demonstrate that astrocyte-mediated synapse elimination is not merely a housekeeping function, but a necessary component of functional circuit refinement during learning.
The team further investigated how astrocytes determine which synapses to remove and identified two major regulatory signals. First, increasing neuronal activity between the motor cortex and the striatum significantly enhanced astrocyte-mediated synaptic elimination (a process in which astrocytes engulf and remove synapses), indicating that circuit engagement promotes remodeling. Second, manipulating dopamine levels, a key neuromodulator for movement and reward, also strongly influenced astrocytic synapse elimination.
Importantly, dopamine produced distinct structural changes in two major types of striatal projection neurons—D1 and D2 medium spiny neurons. Both these changes were found to be dependent on astrocytic MEGF10.
The findings suggest that dopamine helps determine which neurons become more active during learning, while astrocytes reshape the circuit by selectively preserving stronger connections and removing weaker ones. This allows the astrocytes to help translate dopamine signals into lasting structural changes in motor circuits.
By revealing an astrocyte-dependent mechanism underlying dopamine-driven circuit remodeling, the study provides new insight into how motor skills are acquired at the cellular level.
Because dopamine signaling is disrupted in disorders such as Parkinson’s disease and addiction, understanding how astrocytes contribute to dopamine-regulated plasticity may inform future investigations into circuit dysfunction in these conditions.
Associate Diretor CHUNG Won-Suk noted, “Learning depends on a precise circuit rewiring process that involves not only forming new synapses but also removing unnecessary connections. Our study systematically identifies astrocytic phagocytosis and MEGF10 as key players in this process.”
Key Questions Answered:
A: That’s the old view! We now know astrocytes are active players. In this study, they act like the “editors” of a book—while neurons write the sentences, astrocytes delete the unnecessary ones so the “story” of a motor skill makes sense.
A: Think of dopamine as the “highlighting pen.” It highlights the important neural paths, and astrocytes then come along and delete everything that isn’t highlighted. This ensures the brain builds a fast, efficient circuit for the new skill you’re learning.
A: Potentially. Parkinson’s involves a loss of dopamine in the striatum. If we understand how astrocytes and dopamine work together to prune synapses, we might find new ways to treat the movement problems and learning deficits associated with dopamine-related disorders.
Editorial Notes:
- This article was edited by a Neuroscience News editor.
- Journal paper reviewed in full.
- Additional context added by our staff.
About this neuroscience research news
Author: William Suh
Source: Institute for Basic Science
Contact: William Suh – Institute for Basic Science
Image: The image is credited to Neuroscience News
Original Research: Open access.
“Motor learning and dopamine-dependent striatal synaptic plasticity are controlled by astrocytic MEGF10” by Young-Jin Choi, Youngeun Lina Lee, Yemin Kim, Jaeseon Jeon, Jae-Ick Kim & Won-Suk Chung. Nature Communications
DOI:10.1038/s41467-026-69129-1
Abstract
Motor learning and dopamine-dependent striatal synaptic plasticity are controlled by astrocytic MEGF10
Dopamine regulates motor learning by modulating striatal synaptic plasticity in medium spiny neurons (MSNs).
Despite its well-established role in synaptic plasticity, dopamine’s involvement in glia-mediated synapse remodeling remains unclear.
Here, we demonstrate that the astrocytic phagocytic receptor MEGF10 (Multiple Epidermal Growth Factor-like Domains Protein 10), but not MERTK (MER Proto-Oncogene, Tyrosine Kinase), is required for the elimination of corticostriatal excitatory synapses on MSNs during motor learning.
Deletion of astrocytic Megf10 impaired long-term potentiation and depression (LTP and LTD), and reduced learning-induced increases in synaptic strength.
Notably, chemogenetic activation of corticostriatal transmission or dopamine release from the substantia nigra pars compacta (SNc) selectively enhanced astrocytic synapse elimination.
Furthermore, elevated dopamine and motor learning differentially regulated postsynaptic elimination in MSNs depending on dopamine receptor subtype, leading to MEGF10-dependent changes in synaptic remodeling and quantal properties.
These findings identify astrocytic MEGF10 as a key mediator of dopamine- and activity-dependent synapse remodeling in the striatum.