Summary: A new mouse model of Alzheimer’s more closely resembles the human version of the disease. Researchers believe the new model may help accelerate new therapeutic avenues to treat the disease.
Source: University of Pennsylvania.
By injecting human Alzheimer’s disease brain extracts of pathological tau protein (from postmortem donated tissue) into mice with different amounts of amyloid-beta (Aβ) plaques in their brains, researchers from the Perelman School of Medicine at the University of Pennsylvania found that amyloid-beta facilitates the interaction between the plaques and abnormal tau. This relationship promotes the spread of mutated tau proteins in neurons, which is the hallmark of long-term Alzheimer’s disease. They published their findings this week in Nature Medicine.
“Making an AD mouse model that incorporates both Aβ and tau pathologies in a more AD-relevant context has been greatly sought after but difficult to accomplish,” said senior author Virginia M-Y Lee, PhD, director of the Center for Neurodegenerative Disease Research (CNDR) at Penn. “This study is a big step for AD research, which will allow us to test new therapies in a more realistic context.”
Alzheimer’s disease is characterized by Aβ plaques outside cells and clumps of tau within cells. Researchers have proposed that AB plaques are the initiating pathology of AD, but the failure of all AD clinical trials based on removing AB challenges this hypothesis and the idea of targeting Aβ alone to treat AD. At the same time, evidence from other studies, including research from CNDR, strongly correlates the spread of tau clumps with worsening cognition in AD, but the exact link between the two pathologies has remained enigmatic.
Tau works like railroad track crossties in stabilizing microtubules in axons responsible for transporting material inside neurons. Removal of tau protein from microtubules due to its clumping in nerve cells causes the affected neurons to become dysfunctional, ultimately leading to their death and AD.
The Penn team mimicked the formation of three major types of AD-relevant tau pathology in their new mouse model: neurofibrillary tangles, neuropil threads, and tau aggregates surrounding Aβ plaques, called neuritic plaque tau. “For the first time we could see and study the tau clumps in dystrophic axons surrounding AB plaques in a mouse model, just like we see in a human brain with AD,” said first author Zhuohao He, PhD, a postdoctoral fellow in Lee’s lab.
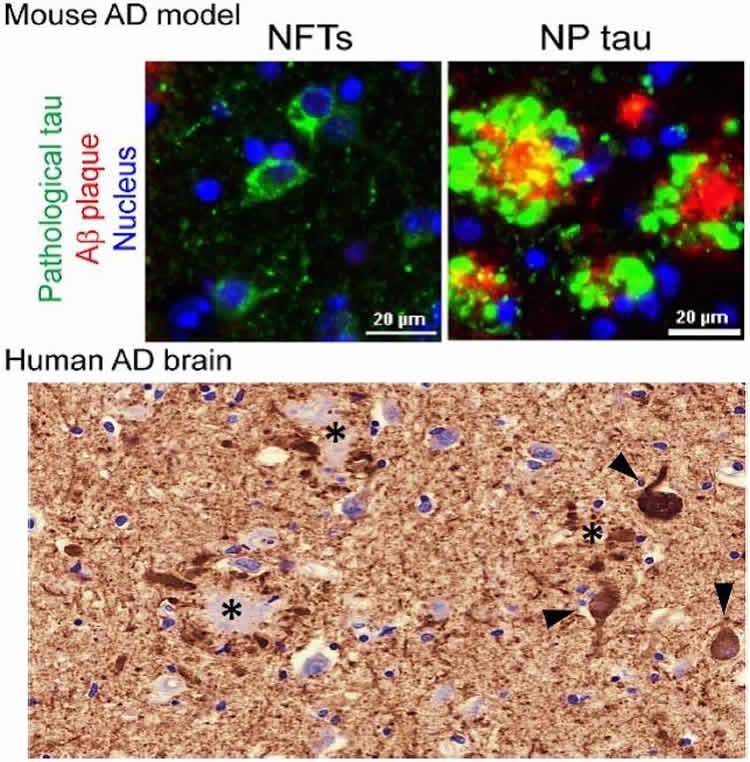
The team found that Aβ plaques create an environment that facilitates the rapid amplification and spread of pathological tau into large aggregates, initially appearing as neuritic plaque tau. This was followed by the formation and spread of neurofibrillary tangles and neuropil threads to other neurons. These tau protein formations also impaired brain functions, including memory difficulties, in the mice.
This study is the basis for a new way to explain how the Aβ plaque environment accelerates the spread of tau pathology in the brains of AD patients, which is consistent with imaging studies and investigations of postmortem AD brains.
The findings suggest new targets and strategies to treat AD patients. “Our new mouse model of AD with both Aβ and tau can now be used to test therapies that target one or both pathologies to see if combination or single-target therapy is better,” Lee said.
Study coauthors are Jing L. Guo, Jennifer D. McBride, Sneha Narasimhan, Hyesung Kim, Lakshmi Changolkar, Bin Zhang, Ronald J. Gathagan, Cuiyong Yue, Christopher Dengler, Anna Stieber, Magdalena Nitla, Douglas A. Coulter, Ted Abel, Kurt R. Brunden, and John Q. Trojanowski.
Funding: This study was funded by the National Institute on Aging, part of the National Institutes of Health (P30AG10124, P01AG17586, P01AG017628).
Source: Karen Kreeger – University of Pennsylvania
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Virginia Lee, PhD, Perelman School of Medicine, University of Pennsylvania.
Original Research: Full open access research for “Amyloid-β plaques enhance Alzheimer’s brain tau-seeded pathologies by facilitating neuritic plaque tau aggregation” by Zhuohao He, Jing L Guo, Jennifer D McBride, Sneha Narasimhan, Hyesung Kim, Lakshmi Changolkar, Bin Zhang, Ronald J Gathagan, Cuiyong Yue, Christopher Dengler, Anna Stieber, Magdalena Nitla, Douglas A Coulter, Ted Abel, Kurt R Brunden, John Q Trojanowski & Virginia M-Y Lee in Nature Medicine. Published online December 4 2017 doi:10.1038/nm.4443
[cbtabs][cbtab title=”MLA”]University of Pennsylvania “New Alzheimer’s Model More Closely Mimics Human Disease.” NeuroscienceNews. NeuroscienceNews, 4 December 2017.
<https://neurosciencenews.com/alzheimers-model-disease-8102/>.[/cbtab][cbtab title=”APA”]University of Pennsylvania (2017, December 4). New Alzheimer’s Model More Closely Mimics Human Disease. NeuroscienceNews. Retrieved December 4, 2017 from https://neurosciencenews.com/alzheimers-model-disease-8102/[/cbtab][cbtab title=”Chicago”]University of Pennsylvania “New Alzheimer’s Model More Closely Mimics Human Disease.” https://neurosciencenews.com/alzheimers-model-disease-8102/ (accessed December 4, 2017).[/cbtab][/cbtabs]
Abstract
Amyloid-β plaques enhance Alzheimer’s brain tau-seeded pathologies by facilitating neuritic plaque tau aggregationy
Alzheimer’s disease (AD) is characterized by extracellular amyloid-β (Aβ) plaques and intracellular tau inclusions. However, the exact mechanistic link between these two AD lesions remains enigmatic. Through injection of human AD-brain-derived pathological tau (AD-tau) into Aβ plaque–bearing mouse models that do not overexpress tau, we recapitulated the formation of three major types of AD-relevant tau pathologies: tau aggregates in dystrophic neurites surrounding Aβ plaques (NP tau), AD-like neurofibrillary tangles (NFTs) and neuropil threads (NTs). These distinct tau pathologies have different temporal onsets and functional consequences on neural activity and behavior. Notably, we found that Aβ plaques created a unique environment that facilitated the rapid amplification of proteopathic AD-tau seeds into large tau aggregates, initially appearing as NP tau, which was followed by the formation and spread of NFTs and NTs, likely through secondary seeding events. Our study provides insights into a new multistep mechanism underlying Aβ plaque–associated tau pathogenesis.
“Amyloid-β plaques enhance Alzheimer’s brain tau-seeded pathologies by facilitating neuritic plaque tau aggregation” by Zhuohao He, Jing L Guo, Jennifer D McBride, Sneha Narasimhan, Hyesung Kim, Lakshmi Changolkar, Bin Zhang, Ronald J Gathagan, Cuiyong Yue, Christopher Dengler, Anna Stieber, Magdalena Nitla, Douglas A Coulter, Ted Abel, Kurt R Brunden, John Q Trojanowski & Virginia M-Y Lee in Nature Medicine. Published online December 4 2017 doi:10.1038/nm.4443






