Summary: Researchers have identified a brain circuit that helps regulate the sleep-wake cycle.
Source: Stanford.
Stanford University School of Medicine scientists have identified a brain circuit that’s indispensable to the sleep-wake cycle. This same circuit is also a key component of the reward system, an archipelago of interconnected brain clusters crucial to promoting behavior necessary for animals, including humans, to survive and reproduce.
It makes intuitive sense that the reward system, which motivates goal-directed behaviors such as fleeing from predators or looking for food, and our sleep-wake cycle would coordinate with one another at some point. You can’t seek food in your sleep, unless you’re an adept sleepwalker. Conversely, getting out of bed is a lot easier when you’re excited about the day ahead of you.
But until this study, no precise anatomical location for this integration of the brain’s reward and arousal systems has been pinpointed, said Luis de Lecea, PhD, professor of psychiatry and behavioral sciences.
The researchers’ findings will be published online Sept. 5 in Nature Neuroscience. De Lecea is the senior author. The lead author is postdoctoral scholar Ada Eban-Rothschild, PhD.
“This has potential huge clinical relevance,” de Lecea said. “Insomnia, a multibillion-dollar market for pharmaceutical companies, has traditionally been treated with drugs such as benzodiazepines that nonspecifically shut down the entire brain. Now we see the possibility of developing therapies that, by narrowly targeting this newly identified circuit, could induce much higher-quality sleep.”
Some 25 to 30 percent of American adults are affected by sleep disturbances of one type or another, according to the National Institutes of Health. In addition, disruption of the sleep-wake rhythm typifies many different neuropsychiatric disorders and is understood to exacerbate them.
One of the first questions a psychiatrist asks a patient, said de Lecea, is, “How’s your sleep?”
Similarity across vertebrates
The reward system’s circuitry is similar in all vertebrates, from fish, frogs and falcons to fishermen and fashion models. A chemical called dopamine plays a crucial role in firing up this circuitry.
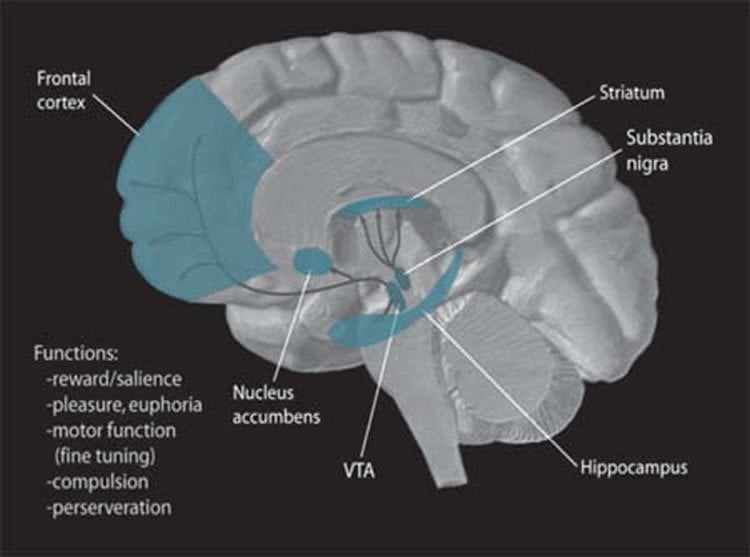
Neuroscientists know that a particular brain structure, the ventral tegmental area, or VTA, is the origin of numerous dopamine-secreting nerve fibers that run in discrete tracts to many different parts of the brain. A plurality of these fibers go to the nucleus accumbens, a forebrain structure particularly implicated in generating feelings of pleasure in anticipation of, or response to, obtaining a desired objective.
“Since many reward-circuit-activating drugs such as amphetamines that work by stimulating dopamine secretion also keep users awake, it’s natural to ask if dopamine plays a key role in the sleep-wake cycle as well as in reward,” Eban-Rothschild said. “But, in part due to existing technical limitations, earlier experimental literature has unearthed little evidence for the connection and, in fact, has suggested that this circuit probably wasn’t so important.”
For the new study, the investigators employed male laboratory mice bioengineered in several respects to enable the use of advanced technologies to remotely excite, suppress and monitor activity in the dopamine-secreting nerve cells from the mice’s VTA. The researchers also measured the mice’s overall brain activity and muscle tone to determine the mice’s relative stages of asleep or arousal. They used video cameras to view the mice’s behavior.
Observed in mice
Overall, activity in the dopamine-secreting nerve cells emanating from the VTA rose on waking and stayed elevated when mice were awake. Conversely, this activity ramped down when mice transitioned into sleep, remaining low while they slumbered. Activating this nerve-cell population was enough to rouse the animals from a sound sleep and keep them awake for long periods, even during a point in the mice’s diurnal cycle when they’d ordinarily be bunking down. Control animals, whose VTA activity wasn’t similarly jacked up, built little nests from pellets of materials placed in all the mice’s cages and then promptly dropped off.
When instead the scientists suppressed activity in the same nerve-cell population during the typically active period of the mice’s 24-hour cycle, the mice conked out, snoozing through the presence of surefire arousal triggers: delicious high-fat chow, a female or fear-inducing fox urine.
Mice in an unfamiliar cage ordinarily explore their new surroundings energetically. And indeed, VTA-suppressed mice stayed awake for the first 45 minutes of the hour they spent in a new cage. But Eban-Rothschild noticed something: They spent that waking time building nests.
“They were really careful about it,” she noted. Once they were satisfied with what they’d built, they dozed off.
This wasn’t just some stereotyped behavior guaranteed to emerge when VTA activity was inhibited, Eban-Rothschild added. “If we put the nest they’d already built in their usual cage into the novel cage, they climbed in and went right to sleep.”
Control mice in the unfamiliar cage ran around, either ignoring the pellet of nesting materials placed inside or scattering those materials all over the cage.
Nest-making activities
Eban-Rothschild analyzed video footage of the animals’ behavior in their novel environments, and correlated 1-second video segments with recorded brain activity during the corresponding time frame. She saw that actions directly connected to building nests were marked by reduced VTA activity, while actions that weren’t were associated with higher levels of VTA activity.
“We knew stimulating the brain’s dopamine-related circuitry would increase goal-directed behaviors such as food- and sex-seeking” said Eban-Rothschild. “But the new study shows that at least one complex behavior is induced not by stimulating, but by inhibiting, this very circuit. Interestingly, this behavior — nest building — is essential to a mouse’s preparation for sleep.”
Nobody had noticed that before, said de Lecea. “This is the first finding of a sleep-preparation starter site in the brain. It’s likely we humans have one, too. If we’re disrupting this preparation by, say, reading email or playing videogames, which not only give off light but charge up our emotions and get our VTA dopaminergic circuitry going, it’s easy to see why we’re likely to have trouble falling asleep.”
Noting that this anticipatory phase is often at the root of many people’s sleeping problems, de Lecea suggested that the newly identified circuit could be a target for pharmacological intervention to help people ease into sleep.
“We have plenty of drugs that counter dopamine,” he said. “Perhaps giving a person the right dose, at just the right time, of a drug with just the right pharmacokinetic properties so its effect will wear off at the right time would work a lot better than bombarding the brain with benzodiazepines, such as Valium, that knock out the entire brain.”
He said he also sees the possibility that drugs targeting the VTA’s dopamine-secreting nerve cells could benefit those suffering from neurological conditions such as schizophrenia or bipolar disorder that are characterized by sleep-wake cycle disturbances.
“It could be that merely solving the sleep-wake part will clear up a lot of symptoms,” de Lecea said.
Source: Bruce Goldman – Stanford
Image Source: This NeuroscienceNews.com image is in the public domain.
Original Research: Abstract for “VTA dopaminergic neurons regulate ethologically relevant sleep–wake behaviors” by Ada Eban-Rothschild, Gideon Rothschild, William J Giardino, Jeff R Jones and Luis de Lecea in Nature Neuroscience. Published online September 5 2016 doi:10.1038/nn.4377
[cbtabs][cbtab title=”MLA”]Stanford. “Brain Circuit That Drives Sleep Wake State and Sleep Preparation Behavior Identified.” NeuroscienceNews. NeuroscienceNews, 5 September 2016.
<https://neurosciencenews.com/sleep-prep-circadian-behavior-4962/>.[/cbtab][cbtab title=”APA”]Stanford. (2016, September 5). Brain Circuit That Drives Sleep Wake State and Sleep Preparation Behavior Identified. NeuroscienceNews. Retrieved September 5, 2016 from https://neurosciencenews.com/sleep-prep-circadian-behavior-4962/[/cbtab][cbtab title=”Chicago”]Stanford. “Brain Circuit That Drives Sleep Wake State and Sleep Preparation Behavior Identified.” https://neurosciencenews.com/sleep-prep-circadian-behavior-4962/ (accessed September 5, 2016).[/cbtab][/cbtabs]
Abstract
Non-invasive monitoring of spreading depression
Dopaminergic ventral tegmental area (VTA) neurons are critically involved in a variety of behaviors that rely on heightened arousal, but whether they directly and causally control the generation and maintenance of wakefulness is unknown. We recorded calcium activity using fiber photometry in freely behaving mice and found arousal-state-dependent alterations in VTA dopaminergic neurons. We used chemogenetic and optogenetic manipulations together with polysomnographic recordings to demonstrate that VTA dopaminergic neurons are necessary for arousal and that their inhibition suppresses wakefulness, even in the face of ethologically relevant salient stimuli. Nevertheless, before inducing sleep, inhibition of VTA dopaminergic neurons promoted goal-directed and sleep-related nesting behavior. Optogenetic stimulation, in contrast, initiated and maintained wakefulness and suppressed sleep and sleep-related nesting behavior. We further found that different projections of VTA dopaminergic neurons differentially modulate arousal. Collectively, our findings uncover a fundamental role for VTA dopaminergic circuitry in the maintenance of the awake state and ethologically relevant sleep-related behaviors.
“VTA dopaminergic neurons regulate ethologically relevant sleep–wake behaviors” by Ada Eban-Rothschild, Gideon Rothschild, William J Giardino, Jeff R Jones and Luis de Lecea in Nature Neuroscience. Published online September 5 2016 doi:10.1038/nn.4377






