Summary: A new study reveals how THC binds to cannabinoid receptor 1 in the brain.
Source: Cell Press.
Researchers have the clearest picture yet of the receptor that causes the “high” associated with marijuana. The three-dimensional image of cannabinoid receptor 1, revealed October 20 in Cell, reveals how molecules like THC bind to cannabinoid receptor 1, which is found to be embedded in the surface of many nerve cells. This could explain how pain medications meant to mimic cannabis use without the “high” can cause unintended side effects and provide a foundation for future therapies.
“With marijuana becoming more popular with legislation in the United States, we need to understand how molecules like THC (the psychoactive component of marijuana) and the synthetic cannabinoids interact with the receptor, especially since we’re starting to see people show up in emergency rooms when they use synthetic cannabinoids,” says co-author Raymond Stevens, Professor at the iHuman Institute, ShanghaiTech University and Provost Professor of Biological Science and Chemistry at the University of Southern California.
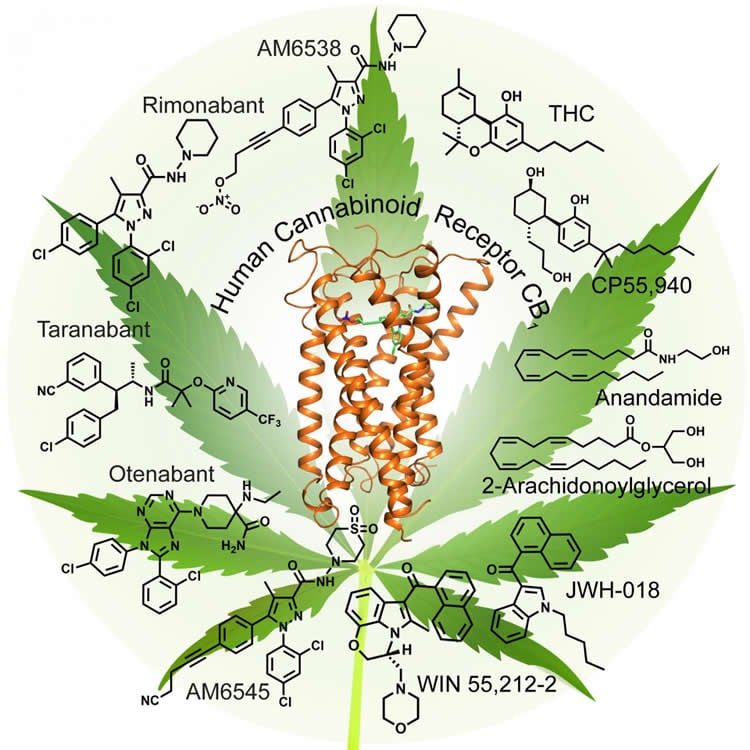
“Researchers are fascinated by how you can make changes in THC or synthetic cannabinoids and have such different effects,” says Stevens. “Now that we finally have the structure of CB1, we can start to understand how these changes to the drug structure can affect the receptor.”
For example, cannabis-derived therapies have been used as a medication for obesity, but, upon investigation, researchers found that the therapeutic molecules could trigger depression, anxiety, and even suicidal tendencies, so the drug was pulled off the market. Currently, researchers are watching for potential side effects related to synthetic cannabinoids, such as Spice and K2, which aim to replicate the “high” associated with THC but instead are triggering trips to the emergency room.
“We need to understand how marijuana works in our bodies; it can have both therapeutic potential and recreational use, but cannabinoids can also be very dangerous,” says iHuman Institute Professor Zhi-jie Liu, co-leader of the study. “By doing both the basic science and understanding how this receptor works, we can then use it to help people in the future.”
Funding: This work was supported through the iHuman Institute at ShanghaiTech University; the National Laboratory of Biomacromolecules at Institute of Biophysics, Chinese Academy of Sciences; the Center for Drug Discovery at Northeastern University, USA; the Departments of Biological Sciences and Chemistry at the University of Southern California; the Departments of Molecular Therapeutics and Neuroscience at the Scripps Research Institute; the University of California, San Diego; the Shanghai Institute of Materia Medica in Shanghai; the GPCR Consortium in San Marcos, California; and the National Institute of Drug Abuse of the National Institutes of Health, USA.
Source: Michaela Kane – Cell Press
Image Source: NeuroscienceNews.com image is credited to Yekaterina Kadyshevskaya, Stevens Laboratory, USC.
Original Research: Full open access research for “Crystal Structure of the Human Cannabinoid Receptor CB1” by Tian Hua, Kiran Vemuri, Mengchen Pu, Lu Qu, Gye Won Han, Yiran Wu, Suwen Zhao, Wenqing Shui, Shanshan Li, Anisha Korde, Robert B. Laprairie, Edward L. Stahl, Jo-Hao Ho, Nikolai Zvonok, Han Zhou, Irina Kufareva, Beili Wu, Qiang Zhao, Michael A. Hanson, Laura M. Bohn, Alexandros Makriyannis, Raymond C. Stevens, Zhi-Jie Liu in Cell. Published online October 20 2016 doi:10.1016/j.cell.2016.10.004
[cbtabs][cbtab title=”MLA”]Cell Press. “High Times: Taking A Look at the Marijuana Receptor.” NeuroscienceNews. NeuroscienceNews, 20 October 2016.
<https://neurosciencenews.com/thc-cannabinoid-receptor-5316/>.[/cbtab][cbtab title=”APA”]Cell Press. (2016, October 20). High Times: Taking A Look at the Marijuana Receptor. NeuroscienceNews. Retrieved October 20, 2016 from https://neurosciencenews.com/thc-cannabinoid-receptor-5316/[/cbtab][cbtab title=”Chicago”]Cell Press. “High Times: Taking A Look at the Marijuana Receptor.” https://neurosciencenews.com/thc-cannabinoid-receptor-5316/ (accessed October 20, 2016).[/cbtab][/cbtabs]
Abstract
Crystal Structure of the Human Cannabinoid Receptor CB1
Highlights
•AM6538 is presented as a stabilizing, tight binding antagonist of CB1
•Crystal structure of human CB1 in complex with AM6538 is determined
•Molecular docking predicts CB1 binding modes of THC and synthetic cannabinoids
•Resolution of the binding pocket provides path for rational CB1 drug design
Summary
Cannabinoid receptor 1 (CB1) is the principal target of Δ9-tetrahydrocannabinol (THC), a psychoactive chemical from Cannabis sativa with a wide range of therapeutic applications and a long history of recreational use. CB1 is activated by endocannabinoids and is a promising therapeutic target for pain management, inflammation, obesity, and substance abuse disorders. Here, we present the 2.8 Å crystal structure of human CB1 in complex with AM6538, a stabilizing antagonist, synthesized and characterized for this structural study. The structure of the CB1-AM6538 complex reveals key features of the receptor and critical interactions for antagonist binding. In combination with functional studies and molecular modeling, the structure provides insight into the binding mode of naturally occurring CB1 ligands, such as THC, and synthetic cannabinoids. This enhances our understanding of the molecular basis for the physiological functions of CB1 and provides new opportunities for the design of next-generation CB1-targeting pharmaceuticals.
“Crystal Structure of the Human Cannabinoid Receptor CB1” by Tian Hua, Kiran Vemuri, Mengchen Pu, Lu Qu, Gye Won Han, Yiran Wu, Suwen Zhao, Wenqing Shui, Shanshan Li, Anisha Korde, Robert B. Laprairie, Edward L. Stahl, Jo-Hao Ho, Nikolai Zvonok, Han Zhou, Irina Kufareva, Beili Wu, Qiang Zhao, Michael A. Hanson, Laura M. Bohn, Alexandros Makriyannis, Raymond C. Stevens, Zhi-Jie Liu in Cell. Published online October 20 2016 doi:10.1016/j.cell.2016.10.004