Summary: Impaired blood flow to specific areas of the brain coincides with Tau buildup in Alzheimer’s patients. As cognition declines, the relationship between vascular dysfunction and Tau accumulation strengthens.
Source: SfN
In Alzheimer’s disease, impaired blood flow to brain regions coincides with tau protein buildup. This relationship strengthens as cognition declines, according to new research published in Journal of Neuroscience.
Vascular function declines and amyloid-β and tau protein accumulate as Alzheimer’s disease progresses, resulting in neuron death. Like the proverbial chicken and egg, it remains unclear if impaired blood flow causes or is caused by errant protein buildup, or if the two symptoms occur for unrelated reasons.
Albrecht et al. used MRI and PET to compare blood flow and tau buildup in the brains of older adults, with cognition ranging from cognitively normal to showing signs of mild cognitive impairment.
Areas with increased tau levels had diminished blood flow, particularly in the inferior temporal gyrus, one of the first regions to show tau buildup in Alzheimer’s disease, even before cognitive symptoms manifest.
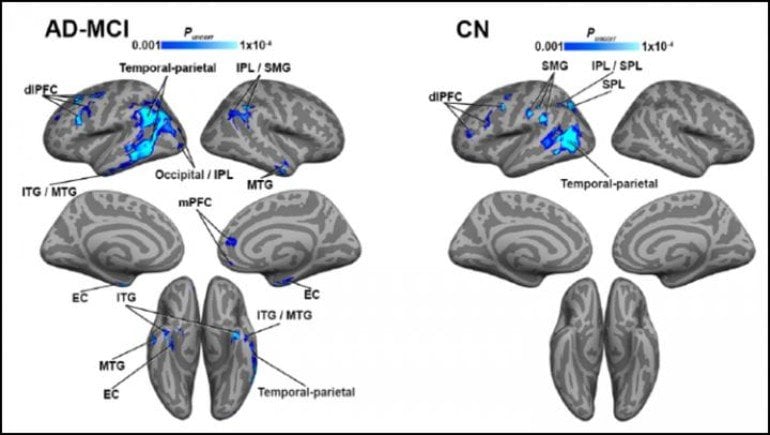
The relationship held true for a separate data pool from the Alzheimer’s Disease Neuroimaging Initiative, which included people with mild cognitive impairment and Alzheimer’s dementia. The correlation between tau and vascular function was stronger in people with greater cognitive impairment and higher amyloid-β levels. It also appeared in more brain regions as the disease progressed in severity.
These findings suggest targeting vascular function could be key to preventing and treating Alzheimer’s dementia.
About this neurology research news
Source: SfN
Contact: Calli McMurray – SfN
Image: The image is credited to Albrecht et al., JNeurosci 2020.
Original Research: Closed access.
“Associations Between Vascular Function and Tau PET are Associated with Global Cognition and Amyloid” by Daniel Albrecht, A. Lisette Isenberg, Joy Stradford, Teresa Monreal, Abhay Sagare, Maricarmen Pachicano, Melanie Sweeney, Arthur Toga, Berislav Zlokovic, Helena Chui, Elizabeth Joe, Lon Schneider, Peter Conti, Kay Jann, Judy Pa and for the Alzheimer Disease Neuroimaging Initiative. Journal of Neuroscience.
Abstract
Associations Between Vascular Function and Tau PET are Associated with Global Cognition and Amyloid
au pathology and vascular dysfunction are important contributors to Alzheimer’s Disease (AD), but vascular-tau associations and their effects on cognition are poorly understood. We investigated these associations in male and female humans by conducting voxelwise comparisons between cerebral blood flow (CBF) and tau positron emission tomography (PET) images in independent discovery (cognitively normal (CN): 19; mild cognitive impairment (MCI)-risk: 43; MCI: 6) and replication (CN: 73; MCI:45; AD: 20) cohorts. In a subgroup, we assessed relationships between tau and soluble platelet-derived growth factor beta (sPDGFRβ), a CSF marker of pericyte injury. We tested whether CBF/sPDGFRβ-tau relationships differed based on Montreal Cognitive Assessment (MoCA) global cognition performance, or based on amyloid burden. Mediation analyses assessed relationships between CBF/sPDGFRβ, tau, and cognition.
Negative CBF-tau correlations were observed predominantly in temporal-parietal regions. In the replication cohort, early negative CBF-tau correlations increased in spatial extent and in strength of correlation with increased disease severity. Stronger CBF- and sPDGFRβ-tau correlations were observed in participants with greater amyloid burden and lower MoCA scores. Importantly, when stratifying by amyloid status, stronger CBF-tau relationships in individuals with lower MoCA scores were driven by amyloid-positive participants. Tau PET was a significant mediator CBF/sPDGFRβ-MoCA relationships in numerous regions.
Our results demonstrate vascular-tau associations across the AD spectrum and suggest that early vascular-tau associations are exacerbated in the presence of amyloid, consistent with a two-hit model of AD on cognition. Combination treatments targeting vascular health, as well as amyloid-β and tau levels, may preserve cognitive function more effectively than single-target therapies.
SIGNIFICANCE
Emerging evidence demonstrates a role for vascular dysfunction as a significant contributor to Alzheimer’s pathophysiology. However, associations between vascular dysfunction and tau pathology, and their effects on cognition remain poorly understood. Multimodal neuroimaging data from two independent cohorts were analyzed to provide novel in vivo evidence of associations between cerebral blood flow (CBF), an MRI measure of vascular health, and tau pathology using PET. CBF-tau associations were related to cognition and driven in part by amyloid burden. Soluble platelet-derived growth factor beta (sPDGFRβ), an independent CSF vascular biomarker, confirmed vascular-tau associations in a subgroup analysis. These results suggest that combination treatments targeting vascular health, amyloid-β, and tau levels, may more effectively preserve cognitive function than single-target therapies.






