Summary: Researchers have identified a pathway near the midbrain where neural messages for taste and pain converge, a new study reports.
Source: University of Oklahoma
University of Oklahoma neuroscientists have found a pathway in the brain where taste and pain intersect in a new study that originally was designed to look at the intersection of taste and food temperature. This study was the first time researchers have shown that taste and pain signals come together in the brain and use the same circuitry. OU neuroscientists received a five-year, $1.6 million National Institutes of Health grant to study this concept.
“We originally aimed to look at how sense of taste works with thermal sensation in this study to better understand how taste is connected to food preferences, health and well-being. Taste is also closely tied to emotion and understanding how the brain processes different tastes is significant on several levels,” said Christian Lemon, principal investigator on the grant and associate professor in the OU Department of Biology, OU College of Arts and Sciences. “What we found was a surprise because temperature signals were converging with taste near the mid-brain, but so were neural messages for taste and pain.”
Lemon and Jinrong Li, OU research associate, used a molecular biology and physiology technique to understand how taste and thermal pathways might be converging with pain. What OU researchers learned from this study is that the neural circuitry carrying signals for aversive tastes also carries a response to pain. This intersection may support a protective function and opens the possibility that taste messages could change how pain signals are transmitted in the brain, but more research is needed.
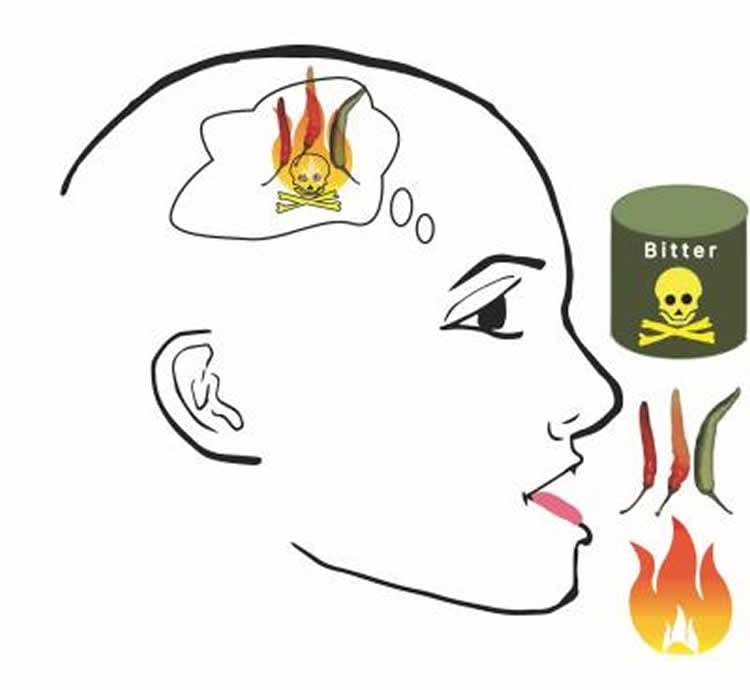
The sense of taste is a complicated sensory and nutrient detector that has many implications for how the nervous system guides food preference behaviors and, potentially, response to pain. Now that the circuitry has been identified, OU researchers will explore manipulation of the circuitry to test its influence on behaviors associated with taste and pain. Ultimately, understanding taste is critical to defining its role in human disorders associated with eating behaviors, such as obesity, diabetes, and other conditions and diseases.
Source:
University of Oklahoma
Media Contacts:
Jana Smith – University of Oklahoma
Image Source:
The image is credited to University of Oklahoma.
Original Research:
“Mouse Parabrachial Neurons Signal a Relationship between Bitter Taste and Nociceptive Stimuli”
Jinrong Li, Christian H. Lemon
Journal of Neuroscience 27 February 2019, 39 (9) 1631-1648; DOI: 10.1523/JNEUROSCI.2000-18.2018
Abstract
Mouse Parabrachial Neurons Signal a Relationship between Bitter Taste and Nociceptive Stimuli
Taste and somatosensation both mediate protective behaviors. Bitter taste guides avoidance of ingestion of toxins while pain sensations, such as noxious heat, signal adverse conditions to ward off harm. Although brain pathways for taste and somatosensation are typically studied independently, prior data suggest that they intersect, potentially reflecting their common protective role. To investigate this, we applied electrophysiologic and optogenetic techniques in anesthetized mice of both sexes to evaluate relationships between oral somatosensory and taste activity in the parabrachial nucleus (PbN), implicated for roles in gustation and pain. Spikes were recorded from taste-active PbN neurons tested with oral delivery of thermal and chemesthetic stimuli, including agonists of nocisensitive transient receptor potential (TRP) ion channels on somatosensory fibers. Gustatory neurons were also tested to follow electrical pulse stimulation of an oral somatosensory region of the spinal trigeminal subnucleus caudalis (Vc), which projects to the PbN. Neurons composed classic taste groups, including sodium, electrolyte, appetitive, or bitter cells. Across groups, most neurons spiked to Vc pulse stimulation, implying that trigeminal projections reach PbN gustatory neurons. Among such cells, a subpopulation responsive to the bitter taste stimuli quinine and cycloheximide, and aversive concentrations of sodium, cofired to agonists of nocisensitive TRP channels, including capsaicin, mustard oil, and noxious heat. Such neurons populated the lateral PbN. Further, nociceptive activity in PbN bitter taste neurons was suppressed during optogenetic-assisted inhibition of the Vc, implying convergent trigeminal input contributed to such activity. Our results reveal a novel role for PbN gustatory cells in cross-system signaling related to protection.
SIGNIFICANCE STATEMENT
Prior data suggest that gustatory and trigeminal neural pathways intersect and overlap in the parabrachial area. However, no study has directly examined such overlap and why it may exist. Here we found that parabrachial gustatory neurons can receive afferent projections from trigeminal nuclei and fire to oral nociceptive stimuli that excite somatosensory receptors and fibers. Activation to aversive nociceptive stimuli in gustatory cells was associated with responding to behaviorally avoided bitter tastants. We were further able to show that silencing trigeminal projections inhibited nociceptive activity in parabrachial bitter taste neurons. Our results imply that in the parabrachial area, there is predictable overlap between taste and somatosensory processing related to protective coding and that classically defined taste neurons contribute to this process.