Researchers at the Max Planck Florida Institute for Neuroscience have uncovered a critical molecule that regulates synaptic transmission.
Facts:
- Neurons communicate with each other through specialized structures called synapses.
- The information is transmitted in the form of synaptic vesicles that contain specific chemical messengers called neurotransmitters
- The amount and coordinated release of neurotransmitters regulates synaptic strength which is critical to maintain proper communication between neurons.
- To better understand and address a number of neurological disorders, we need a better understanding of the molecular mechanisms that regulate neuronal communication.
- A new study has revealed an important function of a class of presynaptic proteins previously implicated in neurological disorders in the regulation of synaptic strength.
Synaptic proteins and neuronal transmission
A synapse consists of a presynaptic terminal of one neuron and a postsynaptic terminal of another. The presynaptic terminal stores vesicles containing neurotransmitters, while the postsynaptic terminal contains neurotransmitter receptors. A dense collection of proteins is present in these terminals, however the functional role of many of these proteins remains unknown.
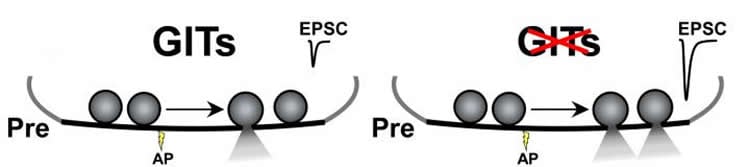
In particular, the G-protein-coupled receptor kinase-interacting proteins (GITs) exert a critical control in synaptic transmission, since deletions of these proteins are lethal or cause sensory deficits and cognitive impairments in mice. In particular, GIT proteins and the pathways they regulate have been implicated in neurological disorders such as Attention Deficit Hyperactivity Disorder (ADHD) and Huntington’s Disease. Several studies have demonstrated the role of GITs in the postsynaptic terminal, but very little is known about their role in the presynaptic terminal. Researchers in Samuel Young Jr.’s research team at the Max Planck Florida Institute for Neuroscience set out to investigate the role of GITs in the giant synapse, the calyx of Held, of the auditory system – the optimal model to study the presynaptic terminal independently from the postsynaptic terminal.
New findings
In their December publication in Neuron, Drs. Samuel Young Jr. and Mónica S. Montesinos and collaborators report for the first time that GIT proteins are critical presynaptic regulators of synaptic strength. This study uncovers previously unknown distinct roles for GIT1 and GIT2 in regulating neurotransmitter release strength, with GIT1 as a specific regulator of presynaptic release probability. This regulation is likely to contribute to the disruptions in neural circuit functions leading to sensory disorders, memory and learning impairment and other neurological disorders.
Future Directions
Future studies of Dr. Samuel Young Jr.’s lab will resolve the mechanisms by which GITs regulate synaptic strength and their roles in the early stages of auditory processing and neurological diseases. “Our work brings significant insight into the understanding of how neuronal communication is regulated, which is essential to understand the cellular and molecular mechanisms of information processing by neuronal circuits and the role of these proteins in the development of neurological diseases,” explained Dr. Young.
Funding: The study was funded by Max Planck Society.
Source: Jennifer Gutierrez – Max Planck Florida Institute for Neuroscience
Image Credit: The image is credited to Mónica S. Montesinos and Samuel M. Young Jr./Max Planck Florida Institute for Neuroscience
Original Research: Abstract for “Presynaptic Deletion of GIT Proteins Results in Increased Synaptic Strength at a Mammalian Central Synapse” by Mónica S. Montesinos, Wei Dong, Kevin Goff, Brati Das, Debbie Guerrero-Given, Robert Schmalzigaug, Richard T. Premont, Rachel Satterfield, Naomi Kamasawa, and Samuel M. Young Jr. in Neuron. Published online December 2 2015 doi:10.1016/j.neuron.2015.10.042
Abstract
Presynaptic Deletion of GIT Proteins Results in Increased Synaptic Strength at a Mammalian Central Synapse
Highlights
•Presynaptic deletion of GITs increases AP-evoked release with no change in RRP size
•Loss of GIT proteins does not affect voltage gated calcium currents
•GIT1 has a specific role in regulating release probability, distinct from GIT2
•GITs regulate synaptic strength by regulation of exocytosis efficiency
Summary
A cytomatrix of proteins at the presynaptic active zone (CAZ) controls the strength and speed of neurotransmitter release at synapses in response to action potentials. However, the functional role of many CAZ proteins and their respective isoforms remains unresolved. Here, we demonstrate that presynaptic deletion of the two G protein-coupled receptor kinase-interacting proteins (GITs), GIT1 and GIT2, at the mouse calyx of Held leads to a large increase in AP-evoked release with no change in the readily releasable pool size. Selective presynaptic GIT1 ablation identified a GIT1-specific role in regulating release probability that was largely responsible for increased synaptic strength. Increased synaptic strength was not due to changes in voltage-gated calcium channel currents or activation kinetics. Quantitative electron microscopy revealed unaltered ultrastructural parameters. Thus, our data uncover distinct roles for GIT1 and GIT2 in regulating neurotransmitter release strength, with GIT1 as a specific regulator of presynaptic release probability.
“Presynaptic Deletion of GIT Proteins Results in Increased Synaptic Strength at a Mammalian Central Synapse” by Mónica S. Montesinos, Wei Dong, Kevin Goff, Brati Das, Debbie Guerrero-Given, Robert Schmalzigaug, Richard T. Premont, Rachel Satterfield, Naomi Kamasawa, and Samuel M. Young Jr. in Neuron. Published online December 2 2015 doi:10.1016/j.neuron.2015.10.042






