Summary: Hyperactivity in the right hemisphere is stronger in people who stutter or have similar speech disorders than in those without speech problems, researchers report.
Source: Max Planck Institute.
One per cent of adults and five per cent of children are unable to achieve what most of us take for granted–speaking fluently. Instead, they struggle with words, often repeating the beginning of a word, for example “G-g-g-g-g-ood morning” or get stuck with single sounds, such as “Ja” for “January” although they know exactly what they want to say.
What processes in the brain cause people to stutter? Previous studies showed imbalanced activity of the two brain hemispheres in people who stutter compared to fluent speakers: A region in the left frontal brain is hypoactive, whereas the corresponding region in the right hemisphere is hyperactive. However, the cause of this imbalance is unclear. Does the less active left hemisphere reflect a dysfunction and causes the right side to compensate for this failure? Or is it the other way around and the hyperactive right hemisphere suppresses activity in the left hemisphere and is therefore the real cause of stuttering?
Scientists at the Max Planck Institute for Human Cognitive and Brain Sciences (MPI CBS) in Leipzig and at the University Medical Center Göttingen have now gained crucial insights: The hyperactivity in regions of the right hemisphere seems to be central for stuttering: “Parts of the right inferior frontal gyrus (IFG) are particularly active when we stop actions, such as hand or speech movements”, says Nicole Neef, neuroscientist at MPI CBS and first author of the new study. “If this region is overactive, it hinders other brain areas that are involved in the initiation and termination of movements. In people who stutter, the brain regions that are responsible for speech movements are particularly affected.”
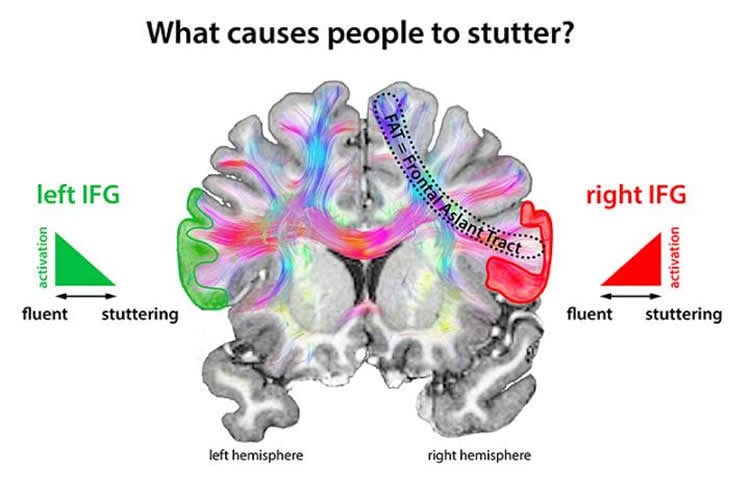
Two of these areas are the left inferior frontal gyrus (IFG), which processes the planning of speech movements, and the left motor cortex, which controls the actual speech movements. “If these two processes are sporadically inhibited, the affected person is unable to speak fluently”, explains Neef.
The scientists investigated these relations using Magnetic Resonance Imaging (MRI) in adults who have stuttered since childhood. In the study, the participants imagined themselves saying the names of the months. They used this method of imaginary speaking to ensure that real speech movements did not interfere with the sensitive MRI signals. The neuroscientists were then able to analyse the brain by scanning for modified fibre tracts in the overactive right hemisphere regions in participants who stutter.
Indeed, they found a fibre tract in the hyperactive right network that was much stronger in affected persons than in those without speech disorders. “The stronger the frontal aslant tract (FAT), the more severe the stuttering. From previous studies we know that this fibre tract plays a crucial role in fine-tuning signals that inhibit movements”, the neuroscientist states. “The hyperactivity in this network and its stronger connections could suggest that one cause of stuttering lies in the neural inhibition of speech movements.”
Source: Verena Müller – Max Planck Institute
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Max Planck Institute for Human Cognitive and Brain Sciences (MPI CBS).
Original Research: Abstract for “Structural connectivity of right frontal hyperactive areas scales with stuttering severity” by Nicole E Neef, Alfred Anwander, Christoph Bütfering, Carsten Schmidt-Samoa, Angela D Friederici, Walter Paulus, and Martin Sommer in Brain. Published online December 8 2017 doi:10.1093/brain/awx316
[cbtabs][cbtab title=”MLA”]Max Planck Institute “Stuttering: Stop Signals in the Brain Disturb Speech Flow.” NeuroscienceNews. NeuroscienceNews, 12 December 2017.
<https://neurosciencenews.com/stuttering-stop-signals-8173/>.[/cbtab][cbtab title=”APA”]Max Planck Institute (2017, December 12). Stuttering: Stop Signals in the Brain Disturb Speech Flow. NeuroscienceNews. Retrieved December 12, 2017 from https://neurosciencenews.com/stuttering-stop-signals-8173/[/cbtab][cbtab title=”Chicago”]Max Planck Institute “Stuttering: Stop Signals in the Brain Disturb Speech Flow.” https://neurosciencenews.com/stuttering-stop-signals-8173/ (accessed December 12, 2017).[/cbtab][/cbtabs]
Abstract
Structural connectivity of right frontal hyperactive areas scales with stuttering severity
A neuronal sign of persistent developmental stuttering is the magnified coactivation of right frontal brain regions during speech production. Whether and how stuttering severity relates to the connection strength of these hyperactive right frontal areas to other brain areas is an open question. Scrutinizing such brain–behaviour and structure–function relationships aims at disentangling suspected underlying neuronal mechanisms of stuttering. Here, we acquired diffusion-weighted and functional images from 31 adults who stutter and 34 matched control participants. Using a newly developed structural connectivity measure, we calculated voxel-wise correlations between connection strength and stuttering severity within tract volumes that originated from functionally hyperactive right frontal regions. Correlation analyses revealed that with increasing speech motor deficits the connection strength increased in the right frontal aslant tract, the right anterior thalamic radiation, and in U-shaped projections underneath the right precentral sulcus. In contrast, with decreasing speech motor deficits connection strength increased in the right uncinate fasciculus. Additional group comparisons of whole-brain white matter skeletons replicated the previously reported reduction of fractional anisotropy in the left and right superior longitudinal fasciculus as well as at the junction of right frontal aslant tract and right superior longitudinal fasciculus in adults who stutter compared to control participants. Overall, our investigation suggests that right fronto-temporal networks play a compensatory role as a fluency enhancing mechanism. In contrast, the increased connection strength within subcortical-cortical pathways may be implied in an overly active global response suppression mechanism in stuttering. Altogether, this combined functional MRI–diffusion tensor imaging study disentangles different networks involved in the neuronal underpinnings of the speech motor deficit in persistent developmental stuttering.
“Structural connectivity of right frontal hyperactive areas scales with stuttering severity” by Nicole E Neef, Alfred Anwander, Christoph Bütfering, Carsten Schmidt-Samoa, Angela D Friederici, Walter Paulus, and Martin Sommer in Brain. Published online December 8 2017 doi:10.1093/brain/awx316






