Summary: A new study repotrs researchers have found evidence as to how muscles may be triggered to regenerate when damaged.
Source: Monash University.
Scientists from Monash University’s Australian Regenerative Medicine Institute ARMI have found the first real evidence of how muscles may be triggered to regenerate or heal when damaged. The research could open the way to improving the lives of the elderly, those with the progressive muscle wasting disease muscular dystrophy, and even enhance muscle recovery in athletes.
In research published in the prestigious journal Science, Professor Peter Currie, Director of ARMI, and his team used the zebra fish to model muscle regeneration.
The small tropical fish are known as “king regenerators” because they have the capacity to regenerate any nerve or muscle that is injured. Zebrafish are also transparent so that scientists are able to observe the regeneration taking place within the living fish.
In the zebra fish model the scientists focused on adult muscle stem cells, which exist in close proximity to mature muscle fibres. When the muscle fibres are injured they send out projections which capture these stem cells, hauling them back in and regenerating the damaged muscle.
While the role of these stem cells in muscle regeneration has long been hypothesized, how they are activated and controlled has only been fully studied in cell samples, and not in a whole animal model. Now this can be studied thanks to the zebrafish.
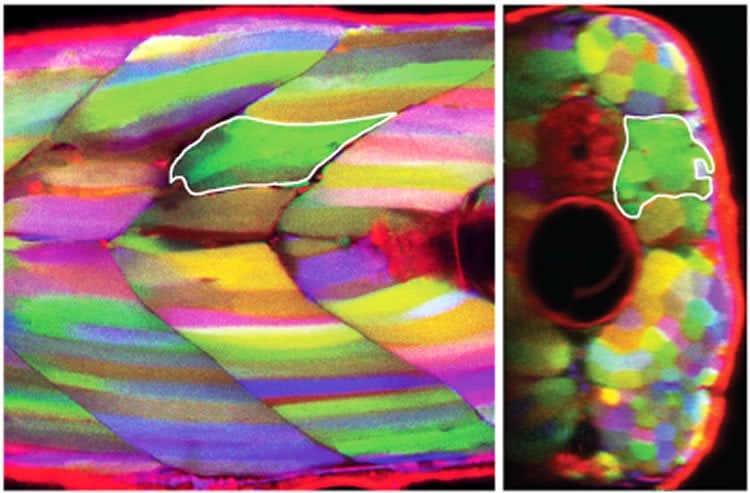
Using specialist microscopy, Professor Currie and his researchers have been able to view and image the regeneration of living muscle tissue in real time, thereby allowing the first glimpse into how these stem cells move, behave and repair the muscle.
Professor Currie has hailed the study as opening the way to learning how to trigger muscle cells in humans to regenerate. “This process has never been caught in action before. It could reveal how we might better stimulate these cells in our own muscles,” he said.
Funding: Funding information not available.
Source: Monash University
Image Source: This NeuroscienceNews.com image is adapted from the Monash University press release.
Original Research: Abstract for “Asymmetric division of clonal muscle stem cells coordinates muscle regeneration in vivo” by David B. Gurevich, Phong Dang Nguyen, Ashley L. Siegel, Ophelia V. Ehrlich, Carmen Sonntag, Jennifer M. N. Phan, Silke Berger, Dhananjani Ratnayake, Lucy Hersey, Joachim Berger, Heather Verkade, Thomas E. Hall, and Peter D. Currie in Science. Published online May 19 2016 doi:10.1126/science.aad9969
[cbtabs][cbtab title=”MLA”]Monash University. “Identifying the First Steps in Muscle Regeneration” NeuroscienceNews. NeuroscienceNews, 20 May 2016.
<https://neurosciencenews.com/stem-cell-muscle-regeneration-4266/>.[/cbtab][cbtab title=”APA”]Monash University. (2016, May 20). Identifying the First Steps in Muscle Regeneration. NeuroscienceNews. Retrieved May 20, 2016 from https://neurosciencenews.com/stem-cell-muscle-regeneration-4266/[/cbtab][cbtab title=”Chicago”]Monash University. “Identifying the First Steps in Muscle Regeneration.” NeuroscienceNews.
https://neurosciencenews.com/stem-cell-muscle-regeneration-4266/ (accessed May 20, 2016).[/cbtab][/cbtabs]
Abstract
Asymmetric division of clonal muscle stem cells coordinates muscle regeneration in vivo
Skeletal muscle is an example of a tissue that deploys a self-renewing stem cell, the satellite cell, to effect regeneration. Recent in vitro studies have highlighted a role for asymmetric divisions in renewing rare “immortal” stem cells and generating a clonal population of differentiation competent myoblasts. This model currently lacks in vivo validation. Here we define a zebrafish muscle stem cell population analogous to the mammalian satellite cell and image the entire process of muscle regeneration from injury to fiber replacement in vivo. This analysis reveals complex interactions between satellite cells and both injured and uninjured fibers and provides in vivo evidence for asymmetric satellite cell division driving both self-renewal and regeneration via a clonally restricted progenitor pool.
“Asymmetric division of clonal muscle stem cells coordinates muscle regeneration in vivo” by David B. Gurevich, Phong Dang Nguyen, Ashley L. Siegel, Ophelia V. Ehrlich, Carmen Sonntag, Jennifer M. N. Phan, Silke Berger, Dhananjani Ratnayake, Lucy Hersey, Joachim Berger, Heather Verkade, Thomas E. Hall, and Peter D. Currie in Science. Published online May 19 2016 doi:10.1126/science.aad9969






