Summary: Combining neuroimaging data with artificial intelligence, researchers have identified two distinct neuroanatomical subtypes of schizophrenia. The first, more typical subtype is associated with a lower widespread volume of gray matter compared to healthy controls. In the second subtype, gray matter volume is largely similar to healthy brains.
Source: University of Pennsylvania
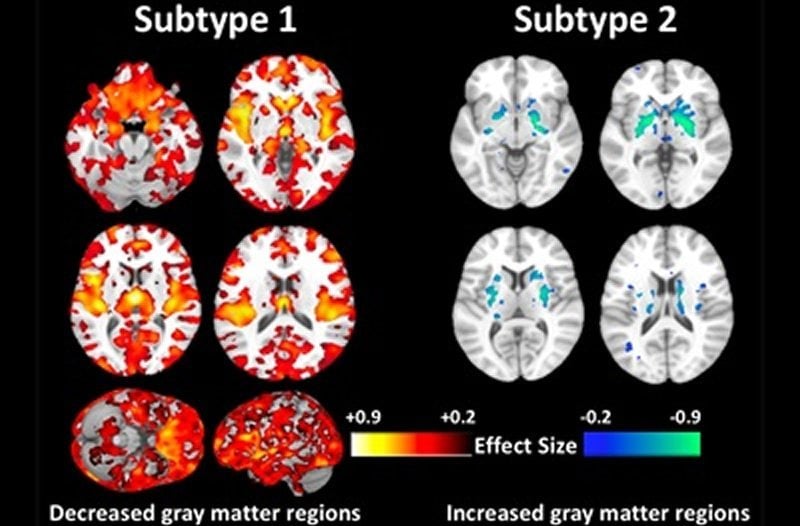
Penn Medicine researchers are the first to discover two distinct neuroanatomical subtypes of schizophrenia after analyzing the brain scans of over 300 patients. The first type showed lower widespread volumes of gray matter when compare to healthy controls, while the second type had volumes largely similar to normal brains. The findings, published Thursday in the journal Brain, suggest that, in the future, accounting for these differences could inform more personalized treatment options.
“Numerous other studies have shown that people with schizophrenia have significantly smaller volumes of brain tissue than healthy controls. However, for at least a third of patients we looked at, this was not the case at all — their brains were almost completely normal,” said principal investigator Christos Davatzikos, PhD, the Wallace T. Miller Professor of Radiology in the Perelman School of Medicine at the University of Pennsylvania. “In the future, we’re not going to be saying, ‘This patient has schizophrenia,’ We’re going to be saying, ‘This patient has this subtype’ or ‘this abnormal pattern,’ rather than having a wide umbrella under which everyone is categorized.”
Schizophrenia is a poorly understood mental disorder that typically presents with hallucinations, delusions, and other cognitive issues — though symptoms and responses to treatment vary widely from patient to patient. Up until now, attempts to study the disease, by comparing healthy to diseased brains, has neglected to account for this heterogeneity, which Davatzikos says has muddled research findings and undermined clinical care.
To better characterize the distinct brain differences within the schizophrenia patient population, Davatzikos established a research consortium that spanned three continents — the United States, China, and Germany. The international cohort of study participants included 307 schizophrenia patients and 364 healthy controls, all of whom were 45-years-old or younger.
Davatzikos and engineering colleagues then analyzed the brain scans using a machine learning method developed at Penn called HYDRA (Heterogeneity Through Discriminative Analysis). The approach helps to identify “true disease subtypes” by limiting the influence of confounding variables, such as age, sex, imaging protocols, and other factors, according to the study authors.
“This method enabled us to sub-categorize patients and find how they differed from the controls, while allowing us, at the same time, to dissect this heterogeneity and tease out multiple pathologies, rather than trying to find a dominant pattern,” Davatzikos said.
After applying this machine learning method to the brain images, the researchers found that 115 patients with schizophrenia, or nearly 40 percent, did not have the typical pattern of reduced gray matter volume that has been historically linked to the disorder. In fact, their brains showed increases of brain volume in the middle of the brain, in an area called the striatum, which plays a role in voluntary movement. When controlling for differences in medication, age, and other demographics, the researchers could not find any clear explanation for the variation.
“The subtype 2 patients are very interesting, because they have similar demographic and clinical measures with subtype 1, and the only differences were their brain structures,” said Ganesh Chand, PhD, a lead author and postdoctoral researcher in the radiology department at Penn.
There are a variety of antipsychotic medications available to manage the symptoms of schizophrenia, but how they will affect a particular patient — both positively or negatively — is often a shot in the dark, according to study co-senior author Daniel Wolf, MD, PhD, an associate professor of Psychiatry at Penn.
“The treatments for schizophrenia work really well in a minority of people, pretty well in most people, and hardly at all in a minority of people. We mostly can’t predict that outcome, so it becomes a matter of trial and error,” Wolf said. “Now that we are starting to understand the biology behind this disorder, then we will hopefully one day have more informed, personalized approaches to treatment.”
As to why an entire subset of patients with schizophrenia have brains that resemble healthy people, Davatzikos is not willing to speculate.
“This is where we are puzzled right now,” Davatzikos said. “We don’t know. What we do know is that
studies that are putting all schizophrenia patients in one group, when seeking associations with response to treatment or clinical measures, might not be using the best approach.”
Future research, he said, will provide a more detailed picture of these subtypes in relation to other aspects of brain structure and function, clinical symptoms, disease progression, and etiology.
Additional Penn authors include: Guray Erus, Aristedidis Sotiras, Erdem Varol, Dhivya Srinivasan, Jimit Doshi, Raymond Pomponio, Taki Shinohara, Ruben C. Gur, Raquel E. Gur, Russell T. Shinohara, Haochang Shou, Yong Fan, and Theodore D. Satterthwaite.
Funding: This research was funded by the National Institutes of Health grant R01MH112070 and by the PRONIA project as funded by the European Union 7th Framework Program grant 602152.
Source:
University of Pennsylvania
Media Contacts:
Lauren Ingeno – University of Pennsylvania
Image Source:
The image is credited to Christos Davatzikos et al..
Original Research: Closed access
“Two distinct neuroanatomical subtypes of schizophrenia revealed using machine learning”. Christos Davatzikos et al.
Brain doi:10.1093/brain/awaa025.
Abstract
Two distinct neuroanatomical subtypes of schizophrenia revealed using machine learning
Neurobiological heterogeneity in schizophrenia is poorly understood and confounds current analyses. We investigated neuroanatomical subtypes in a multi-institutional multi-ethnic cohort, using novel semi-supervised machine learning methods designed to discover patterns associated with disease rather than normal anatomical variation. Structural MRI and clinical measures in established schizophrenia (n = 307) and healthy controls (n = 364) were analysed across three sites of PHENOM (Psychosis Heterogeneity Evaluated via Dimensional Neuroimaging) consortium. Regional volumetric measures of grey matter, white matter, and CSF were used to identify distinct and reproducible neuroanatomical subtypes of schizophrenia. Two distinct neuroanatomical subtypes were found. Subtype 1 showed widespread lower grey matter volumes, most prominent in thalamus, nucleus accumbens, medial temporal, medial prefrontal/frontal and insular cortices. Subtype 2 showed increased volume in the basal ganglia and internal capsule, and otherwise normal brain volumes. Grey matter volume correlated negatively with illness duration in Subtype 1 (r = −0.201, P = 0.016) but not in Subtype 2 (r = −0.045, P = 0.652), potentially indicating different underlying neuropathological processes. The subtypes did not differ in age (t = −1.603, d.f. = 305, P = 0.109), sex (chi-square = 0.013, d.f. = 1, P = 0.910), illness duration (t = −0.167, d.f. = 277, P = 0.868), antipsychotic dose (t = −0.439, d.f. = 210, P = 0.521), age of illness onset (t = −1.355, d.f. = 277, P = 0.177), positive symptoms (t = 0.249, d.f. = 289, P = 0.803), negative symptoms (t = 0.151, d.f. = 289, P = 0.879), or antipsychotic type (chi-square = 6.670, df = 3, P = 0.083). Subtype 1 had lower educational attainment than Subtype 2 (chi-square = 6.389, d.f. = 2, P = 0.041). In conclusion, we discovered two distinct and highly reproducible neuroanatomical subtypes. Subtype 1 displayed widespread volume reduction correlating with illness duration, and worse premorbid functioning. Subtype 2 had normal and stable anatomy, except for larger basal ganglia and internal capsule, not explained by antipsychotic dose. These subtypes challenge the notion that brain volume loss is a general feature of schizophrenia and suggest differential aetiologies. They can facilitate strategies for clinical trial enrichment and stratification, and precision diagnostics.






