The surprise discovery offers a new understanding of Alzheimer’s disease.
Scientists at The Scripps Research Institute (TSRI) have found diverse genomic changes in single neurons from the brains of Alzheimer’s patients, pointing to an unexpected factor that may underpin the most common form of the disease.
A new study, published February 4, 2015 in the online journal eLife, shows that Alzheimer’s brains commonly have many neurons with significantly more DNA and genomic copies of the Alzheimer’s-linked gene, APP, than normal brains.
“Our findings open a new window into the normal and diseased brain by providing the first evidence that DNA variation in individual neurons could be related to brain function and Alzheimer’s disease,” said Jerold Chun, professor at TSRI and its Dorris Neuroscience Center and senior author of the new study.
A Devastating Disease of Unknown Cause
Alzheimer’s disease is an irreversible brain disease that tends to strike older people. It is progressive—impairing memory, destroying motor skills and eventually causing death. The U.S. Centers for Disease Control and Prevention estimates 5.3 million Americans currently have Alzheimer’s disease, and that number is expected to double by 2050 as the population ages.
Scientists still do not know what triggers the majority of Alzheimer’s cases, making it difficult to develop a treatment. Some genes have been identified in families, however 95 percent of cases are “sporadic,” with no link to a gene or family history of Alzheimer’s.
Researchers have long known about disease-related protein accumulations (called amyloid plaques) in the brains of Alzheimer’s patients. They’ve also known that chromosome 21 plays a role in the disease, due to Alzheimer’s-like symptoms in people with Down syndrome (with three copies of chromosome 21). This chromosome contains the APP gene, which can lead to production of the primary component of the damaging amyloid plaques.
Chun and his laboratory group have had a longstanding interest in genomic variation among brain cells, which produces “genomic mosaicism.” In 2001, Chun was the first to report that the brain contains many distinct genomes within its cells—much like the colorful tiles in an artist’s mosaic.
“When we started, genomic mosaicism in the brain was not recognized,” said Chun. “But it turns out there is a remarkable range of genomic changes encompassed by DNA content variation in single brain cells.”
The Alzheimer’s Brain as a Mosaic
In the new study, Chun and his colleagues first set out to analyze the overall DNA content in cells, comparing 32 post-mortem Alzheimer’s brains and 21 post-mortem non-diseased brains.
Remarkably, the researchers found that more than 90 percent of sporadic Alzheimer’s disease brains displayed highly significant DNA increases of hundreds of millions more DNA base-pairs, compared with control samples, showing that genomic mosaicism was altered in the Alzheimer’s brain. Interestingly, these changes were not found everywhere, but were greatest in a part of the brain involved with complex thought.
Next, the researchers used a technique called single-cell qPCR to determine the numbers of APP copies in 154 individual neurons from Alzheimer’s and normal brains. They also tested the neurons using a technique called FISH as an independent method to assess APP copies using fluorescent probes. The researchers then quantified the copies of APP using both techniques.
The tests showed that neurons from patients with sporadic Alzheimer’s disease were four times as likely to contain more than the normal two copies of APP, with some Alzheimer’s neurons containing up to 12 copies of APP, a phenomenon never seen in the controls.
“A lot of people are still not aware of genomic mosaicism in the brain, so to be able to connect it with a disease is really interesting,” said Gwen Kaeser, a graduate student studying in Chun’s lab and co-first author of the study with former graduate student Diane Bushman.
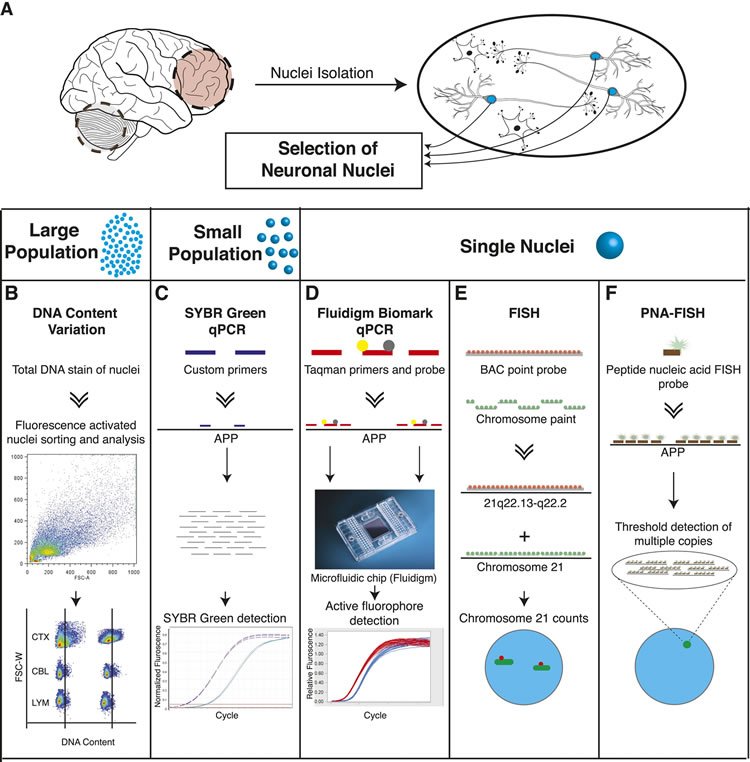
(A) Neuronal nuclei were isolated from the prefrontal cortex and cerebellum of postmortem human brain (see ‘Materials and methods’ for samples used) as described (Westra et al., 2008). (B) Nuclear DNA was stained with propidium iodide (PI) and DNA content was quantified using flow cytometric analysis. (C) APP copy number variations were analyzed in small populations of nuclei (∼75 genomes) using custom primers for exon 14 of APP. (D) Single-cell qPCR assessed APP copy number variations in individual neuronal nuclei via TaqMan probes and a modified Biomark integrated fluidic chip system (Fluidigm Corporation, South San Francisco). (E) FISH paints against the whole q arm of chromosome 21 and a point probe against a region on the q arm of 21 (21q22.13-q22.2) were used to double-label and call aneusomies in AD samples. (F) Peptide nucleic acid (PNA) FISH was combined with super resolution microscopy for threshold detection of APP copy number above ∼2 occurring at a single locus.
A Clue to Other Brain Diseases
While genetic tests on blood samples can reveal if a person is prone to developing an inherited, familial form of Alzheimer’s, most people who develop Alzheimer’s do not test positive. The new study suggests that such genetic signatures might be lacking in the blood of the majority of patients because the genomic signatures of sporadic Alzheimer’s disease occur within individual brain cells.
Indeed, a majority of major brain diseases are also sporadic. For example, amyotrophic lateral sclerosis (ALS) can be linked to a gene in one to two percent of cases, but the rest are sporadic, having no known cause. Chun believes genomic mosaicism could possibly have a role in other brain diseases.
Future studies in the Chun lab will investigate the relationship between mosaicism and disease, the causes of mosaicism and potential new disease drug targets present in the millions of extra base-pairs found in single, Alzheimer’s disease neurons.
In addition to Chun, Kaeser and Bushman, other authors of the study, “Genomic mosaicism with increased amyloid precursor protein (APP) gene copy number in single neurons from sporadic Alzheimer’s disease brains,” were Jurgen W. Westra of TSRI, now at Genedata Inc.; Stevens K. Rehen of TSRI, now at the Federal University of Brazil; and Richard R. Rivera, Benjamin Siddoway and Yun C. Yung, all of TSRI. For more information, see http://dx.doi.org/10.7554/eLife.05116.
Funding: This study received support from the National Institutes of Health (MH076145 and T32 DA07315, T32 AG 000216) and the Shaffer Family Foundation.
Contact: Madeline McCurry-Schmidt – The Scripps Research Institute (TSRI)
Source: The Scripps Research Institute (TSRI) press release
Image Source: The image is from the eLife research article listed below.
Original Research: Full open access research for “Genomic mosaicism with increased amyloid precursor protein (APP) gene copy number in single neurons from sporadic Alzheimer’s disease brains” by Diane M Bushman, Gwendolyn E Kaeser, Benjamin Siddoway, Jurgen W Westra, Richard R Rivera, Stevens K Rehen, Yun C Yung, Jerold Chun in eLife. Published on February 4, 2015. doi: 10.7554/eLife.05116
Abstract: Previous reports have shown that individual neurons of the brain can display somatic genomic mosaicism of unknown function. In this study, we report altered genomic mosaicism in single, sporadic Alzheimer’s disease (AD) neurons characterized by increases in DNA content and amyloid precursor protein (APP) gene copy number. AD cortical nuclei displayed large variability with average DNA content increases of ∼8% over non-diseased controls that were unrelated to trisomy 21. Two independent single-cell copy number analyses identified amplifications at the APP locus. The use of single-cell qPCR identified up to 12 copies of APP in sampled neurons. Peptide nucleic acid (PNA) probes targeting APP, combined with super-resolution microscopy detected primarily single fluorescent signals of variable intensity that paralleled single-cell qPCR analyses. These data identify somatic genomic changes in single neurons, affecting known and unknown loci, which are increased in sporadic AD, and further indicate functionality for genomic mosaicism in the CNS.






