Summary: A new study sheds light on a potential mechanism for the development of schizophrenia. Researchers believe blood vessel abnormalities could play a significant role in the development of the psychiatric disorder.
Source: Trinity College Dublin.
Scientists from Trinity College Dublin and the Royal College of Surgeons in Ireland (RCSI) have discovered that abnormalities of blood vessels in the brain may play a major role in the development of schizophrenia, a debilitating condition that affects around 1% of people in Ireland.
The network of blood vessels in the brain regulates the transport of energy and materials in and out of the brain — forming what is known as the blood-brain barrier (BBB). Scientists working in the Smurfit Institute of Genetics at Trinity College Dublin and the Department of Psychiatry, RCSI, have discovered that abnormalities in the integrity of the BBB may be a critical component in the development of schizophrenia and other brain disorders.
The research, published today in the leading international journal Molecular Psychiatry, was supported by the Health Research Board (HRB), Science Foundation Ireland (SFI) and the US-based charity, Brightfocus Foundation.
People living with a chromosomal abnormality termed ’22q11 deletion syndrome’ (22q11DS) are 20 times more likely to develop schizophrenia. These people lack approximately 40-60 genes within a small region in one of the pairs of chromosome 22. A gene termed “Claudin-5” is located within this region, and it is changes in the levels of this component of the BBB that are associated with the presence of schizophrenia.
Assistant Professor in Neurovascular Genetics at Trinity, Dr Matthew Campbell, said: “Our recent findings have, for the first time, suggested that schizophrenia is a brain disorder associated with abnormalities of brain blood vessels. The concept of tailoring drugs to regulate and treat abnormal brain blood vessels is a novel treatment strategy and offers great potential to complement existing treatments of this debilitating disease.”

“While it is very well accepted that improving cardiovascular health can reduce the risk of stroke and heart attacks, we now believe that drugs aimed at improving cerebrovascular health may be an additional strategy to treating brain diseases in the future.”
Working with an international group of scientists from Cardiff University, Stanford University and Duke University in addition to screening post-mortem brain samples from the Stanley Medical Research Institute, the scientists are the first to identify a molecular genetic component of the blood brain barrier with the development of schizophrenia.
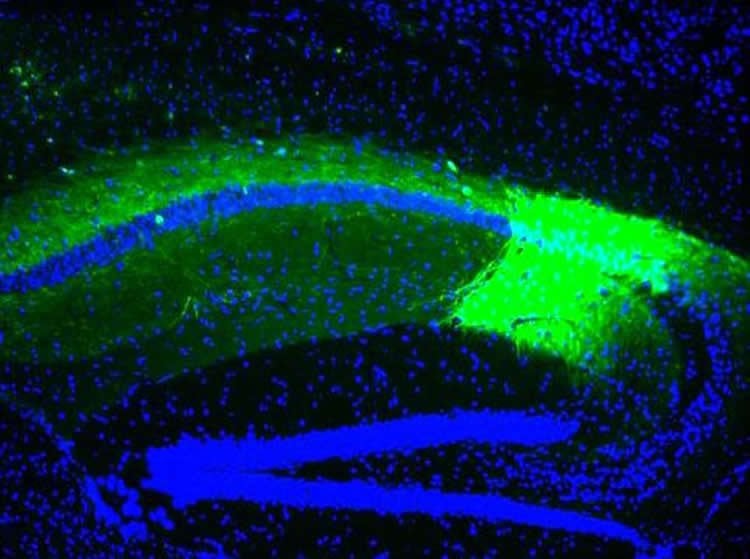
Professor Kieran Murphy, Head of Department of Psychiatry, RCSI and Consultant Psychiatrist at Beaumont Hospital, said: “We have shown for the first time that dysfunction of the blood-brain barrier may be an important factor in the development of schizophrenia. These findings greatly add to our understanding of this debilitating and socially isolating condition.”
Scientists in the laboratories of Dr Matthew Campbell and Professor Kieran Murphy collaborated on this study.
Source: Thomas Deane – Trinity College Dublin
Image Source: NeuroscienceNews.com images are credited to Dr Matthew Campbell, Trinity College Dublin.
Original Research: Full open access research for “Dose-dependent expression of claudin-5 is a modifying factor in schizophrenia” by C Greene, J Kealy, M M Humphries, Y Gong, J Hou, N Hudson, L M Cassidy, R Martiniano, V Shashi, S R Hooper, G A Grant, P F Kenna, K Norris, C K Callaghan, M dN Islam, S M O’Mara, Z Najda, S G Campbell, J S Pachter, J Thomas, N M Williams, P Humphries, K C Murphy & M Campbell in Molecular Psychiatry. Published online October 10 2017 doi:10.1038/mp.2017.156
[cbtabs][cbtab title=”MLA”]Trinity College Dublin “Major Breakthrough Identifies Mechanism for Development of Schizophrenia.” NeuroscienceNews. NeuroscienceNews, 10 October 2017.
<https://neurosciencenews.com/schizophrenia-blood-vessels-7710/>.[/cbtab][cbtab title=”APA”]Trinity College Dublin (2017, October 10). Major Breakthrough Identifies Mechanism for Development of Schizophrenia. NeuroscienceNews. Retrieved October 10, 2017 from https://neurosciencenews.com/schizophrenia-blood-vessels-7710/[/cbtab][cbtab title=”Chicago”]Trinity College Dublin “Major Breakthrough Identifies Mechanism for Development of Schizophrenia.” https://neurosciencenews.com/schizophrenia-blood-vessels-7710/ (accessed October 10, 2017).[/cbtab][/cbtabs]
Abstract
Dose-dependent expression of claudin-5 is a modifying factor in schizophrenia
Schizophrenia is a neurodevelopmental disorder that affects up to 1% of the general population. Various genes show associations with schizophrenia and a very weak nominal association with the tight junction protein, claudin-5, has previously been identified. Claudin-5 is expressed in endothelial cells forming part of the blood-brain barrier (BBB). Furthermore, schizophrenia occurs in 30% of individuals with 22q11 deletion syndrome (22q11DS), a population who are haploinsufficient for the claudin-5 gene. Here, we show that a variant in the claudin-5 gene is weakly associated with schizophrenia in 22q11DS, leading to 75% less claudin-5 being expressed in endothelial cells. We also show that targeted adeno-associated virus-mediated suppression of claudin-5 in the mouse brain results in localized BBB disruption and behavioural changes. Using an inducible ‘knockdown’ mouse model, we further link claudin-5 suppression with psychosis through a distinct behavioural phenotype showing impairments in learning and memory, anxiety-like behaviour and sensorimotor gating. In addition, these animals develop seizures and die after 3–4 weeks of claudin-5 suppression, reinforcing the crucial role of claudin-5 in normal neurological function. Finally, we show that anti-psychotic medications dose-dependently increase claudin-5 expression in vitro and in vivo while aberrant, discontinuous expression of claudin−5 in the brains of schizophrenic patients post mortem was observed compared to age-matched controls. Together, these data suggest that BBB disruption may be a modifying factor in the development of schizophrenia and that drugs directly targeting the BBB may offer new therapeutic opportunities for treating this disorder.
“Dose-dependent expression of claudin-5 is a modifying factor in schizophrenia” by C Greene, J Kealy, M M Humphries, Y Gong, J Hou, N Hudson, L M Cassidy, R Martiniano, V Shashi, S R Hooper, G A Grant, P F Kenna, K Norris, C K Callaghan, M dN Islam, S M O’Mara, Z Najda, S G Campbell, J S Pachter, J Thomas, N M Williams, P Humphries, K C Murphy & M Campbell in Molecular Psychiatry. Published online October 10 2017 doi:10.1038/mp.2017.156






