Summary: Researchers have developed a new probe that can induce blood flow changes that allows for neuroimaging without the use of radioactive labels.
Source: MIT.
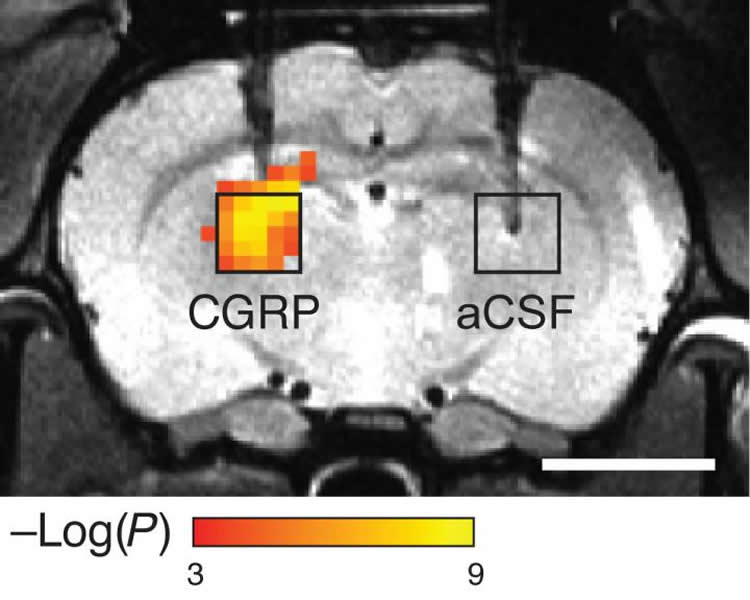
New probe induces blood flow changes that can be seen by MRI.
Scientists hoping to get a glimpse of molecules that control brain activity have devised a new probe that allows them to image these molecules without using any chemical or radioactive labels.
Currently the gold standard approach to imaging molecules in the brain is to tag them with radioactive probes. However, these probes offer low resolution and they can’t easily be used to watch dynamic events, says Alan Jasanoff, an MIT professor of biological engineering.
Jasanoff and his colleagues have developed new sensors consisting of proteins designed to detect a particular target, which causes them to dilate blood vessels in the immediate area. This produces a change in blood flow that can be imaged with magnetic resonance imaging (MRI) or other imaging techniques.
“This is an idea that enables us to detect molecules that are in the brain at biologically low levels, and to do that with these imaging agents or contrast agents that can ultimately be used in humans,” Jasanoff says. “We can also turn them on and off, and that’s really key to trying to detect dynamic processes in the brain.”
In a paper appearing in the December 2 issue of Nature Communications, Jasanoff and his colleagues used these probes to detect enzymes called proteases, but their ultimate goal is to use them to monitor the activity of neurotransmitters, which act as chemical messengers between brain cells.
The paper’s lead authors are postdoc Mitul Desai and former MIT graduate student Adrian Slusarczyk. Recent MIT graduate Ashley Chapin and postdoc Mariya Barch are also authors of the paper.
Indirect imaging
To make their probes, the researchers modified a naturally occurring peptide called calcitonin gene-related peptide (CGRP), which is active primarily during migraines or inflammation. The researchers engineered the peptides so that they are trapped within a protein cage that keeps them from interacting with blood vessels. When the peptides encounter proteases in the brain, the proteases cut the cages open and the CGRP causes nearby blood vessels to dilate. Imaging this dilation with MRI allows the researchers to determine where the proteases were detected.
“These are molecules that aren’t visualized directly, but instead produce changes in the body that can then be visualized very effectively by imaging,” Jasanoff says.
Proteases are sometimes used as biomarkers to diagnose diseases such as cancer and Alzheimer’s disease. However, Jasanoff’s lab used them in this study mainly to demonstrate the validity their approach. Now, they are working on adapting these imaging agents to monitor neurotransmitters, such as dopamine and serotonin, that are critical to cognition and processing emotions.
To do that, the researchers plan to modify the cages surrounding the CGRP so that they can be removed by interaction with a particular neurotransmitter.
“What we want to be able to do is detect levels of neurotransmitter that are 100-fold lower than what we’ve seen so far. We also want to be able to use far less of these molecular imaging agents in organisms. That’s one of the key hurdles to trying to bring this approach into people,” Jasanoff says.
Tracking genes
Another possible application for this type of imaging is to engineer cells so that the gene for CGRP is turned on at the same time that a gene of interest is turned on. That way, scientists could use the CGRP-induced changes in blood flow to track which cells are expressing the target gene, which could help them determine the roles of those cells and genes in different behaviors. Jasanoff’s team demonstrated the feasibility of this approach by showing that implanted cells expressing CGRP could be recognized by imaging.
“Many behaviors involve turning on genes, and you could use this kind of approach to measure where and when the genes are turned on in different parts of the brain,” Jasanoff says.
His lab is also working on ways to deliver the peptides without injecting them, which would require finding a way to get them to pass through the blood-brain barrier. This barrier separates the brain from circulating blood and prevents large molecules from entering the brain.
Funding: The research was funded by the National Institutes of Health BRAIN Initiative and the MIT Simons Center for the Social Brain.
Source: Sarah McDonnell – MIT
Image Source: NeuroscienceNews.com image is credited to the researchers.
Original Research: Full open access research for “Molecular imaging with engineered physiology” by Mitul Desai, Adrian L. Slusarczyk, Ashley Chapin, Mariya Barch & Alan Jasanoff in Nature Communications. Published online December 2 2026 doi:10.1038/ncomms13607
[cbtabs][cbtab title=”MLA”]MIT. “A Radiation-Free Approach to Imaging Molecules in the Brain.” NeuroscienceNews. NeuroscienceNews, 2 December 2026.
<https://neurosciencenews.com/radiation-free-neuroimaging-5659/>.[/cbtab][cbtab title=”APA”]MIT. (2026, December 2). A Radiation-Free Approach to Imaging Molecules in the Brain. NeuroscienceNews. Retrieved December 2, 2026 from https://neurosciencenews.com/radiation-free-neuroimaging-5659/[/cbtab][cbtab title=”Chicago”]MIT. “A Radiation-Free Approach to Imaging Molecules in the Brain.” https://neurosciencenews.com/radiation-free-neuroimaging-5659/ (accessed December 2, 2026).[/cbtab][/cbtabs]
Abstract
Molecular imaging with engineered physiology
In vivo imaging techniques are powerful tools for evaluating biological systems. Relating image signals to precise molecular phenomena can be challenging, however, due to limitations of the existing optical, magnetic and radioactive imaging probe mechanisms. Here we demonstrate a concept for molecular imaging which bypasses the need for conventional imaging agents by perturbing the endogenous multimodal contrast provided by the vasculature. Variants of the calcitonin gene-related peptide artificially activate vasodilation pathways in rat brain and induce contrast changes that are readily measured by optical and magnetic resonance imaging. CGRP-based agents induce effects at nanomolar concentrations in deep tissue and can be engineered into switchable analyte-dependent forms and genetically encoded reporters suitable for molecular imaging or cell tracking. Such artificially engineered physiological changes, therefore, provide a highly versatile means for sensitive analysis of molecular events in living organisms.
“Molecular imaging with engineered physiology” by Mitul Desai, Adrian L. Slusarczyk, Ashley Chapin, Mariya Barch & Alan Jasanoff in Nature Communications. Published online December 2 2026 doi:10.1038/ncomms13607






