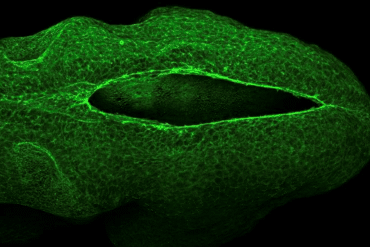
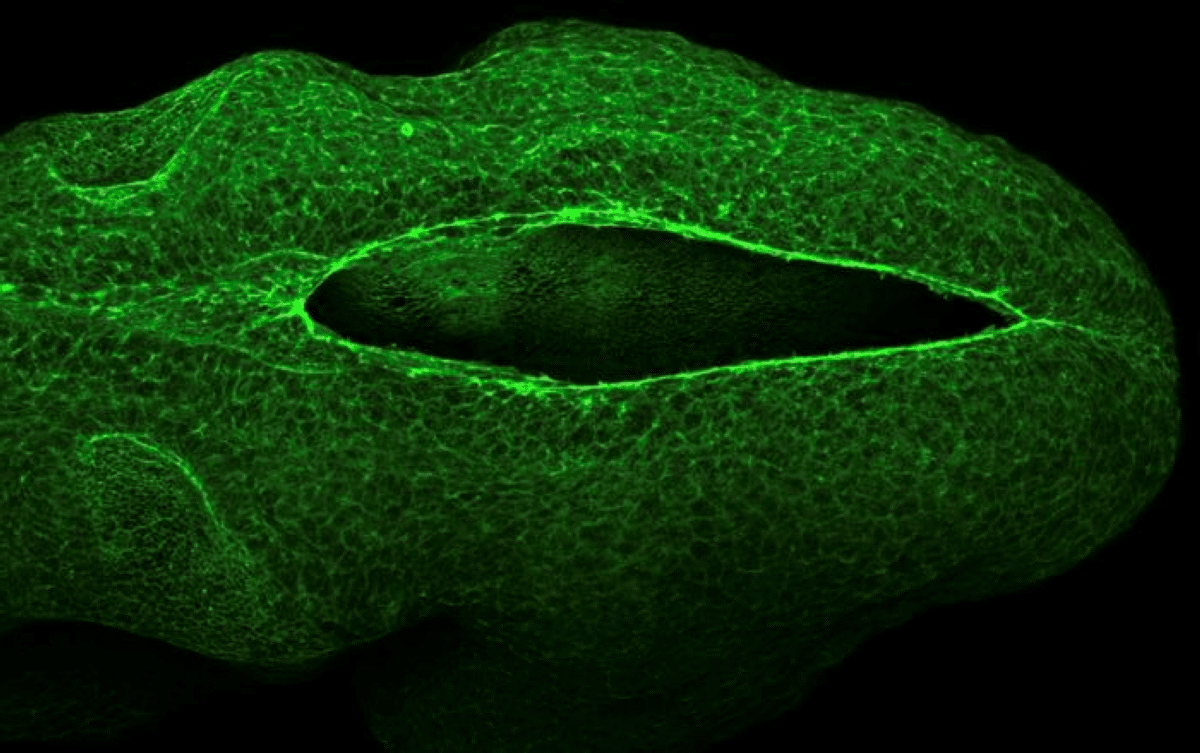
Summary: In a unique collaboration, researchers have decoded the mechanical forces that close the neural tube during the earliest stages of pregnancy. The study reveals that the neural tube doesn’t just “grow” shut; it is physically pulled closed by a “purse-string” mechanism powered by cellular motors.
This breakthrough provides a quantitative framework for understanding why this process fails in one out of every 1,000 pregnancies, leading to severe birth defects like spina bifida.
Key Findings
- Interdisciplinary Breakthrough: By combining theoretical physics with biological imaging, the team “connected the dots” between developmental stages that were previously seen as separate events.
- Mechanical Origins of Birth Defects: The research suggests that neural tube defects may be caused by a mechanical failure in the “purse-string” tension or a lack of cellular coordination, rather than just genetic or nutritional factors alone.
- Beyond the Brain: This physics-based approach to biology can now be applied to other human development stages where force, motion, and timing are critical, such as heart formation or wound healing.
Source: Georgia Tech
In about one out of every 1,000 pregnancies, the neural tube, a key nervous system structure, fails to close properly. Georgia Tech physicists are now helping explain why this happens, having uncovered the physics that drive neural tube closure in a pregnancy’s earliest stages.
Working with collaborators at University College London (UCL), Georgia Tech researchers used computer models to reveal how, during early development, forces generated by cells physically pull the neural tube closed — like a drawstring. This discovery offers new insight into a critical process that, when disrupted, can result in severe birth defects such as spina bifida.
“Understanding a complex developmental process like neural tube closure requires a highly interdisciplinary approach,” said Shiladitya Banerjee, an associate professor in the School of Physics.
“By combining advanced biological imaging with theoretical physics, we were able to uncover the mechanical rules that drive cells to close the tube. My lab builds computational models to uncover the physical rules of living systems. The neural tube is an ideal focus because its formation requires incredible mechanical coordination.”
The researchers presented their findings in Current Biology.
Closing the Gap
The UCL team studied mouse embryos, which develop similarly to humans, and Georgia Tech researchers used that data to construct their models. From the data, they identified the fundamental physics mechanism that enables neural tube closure in part of the brain. This mechanism, called a “purse string,” is made of actin, a pivotal protein that forms a cell’s skeletal structure. As the purse strings tighten, the tube closes.
“These actin molecules are very important because they give rigidity and shape to cells,” Banerjee said.
“During neural tube closure, actin filaments form a ring around the opening and engage molecular motors — proteins that generate forces inside cells,” he said. “As these motors pull on the actin, they generate tension that tightens the ring and draws the tube closed.”
Stretching to Fit
As the actin ring tightens, cells stretch and elongate, causing them to align and move together in a synchronized pattern, like a school of fish. This coordination allows the cells to move faster and more efficiently, increasing tension and driving a feedback loop that helps seal the neural tube.
The team built a computer model to show how this feedback loop leads to successful neural tube formation. Further research using the model could help explain why the neural tube fails to close.
“Physics-based modeling of cell and tissue mechanics allows us to connect the dots between developmental stages in a way that is both robust and quantitative, simulating experiments that are impossible in biological tissues,” said Gabriel Galea, the study co-author and UCL group leader.
“In this case, it allowed us to explain how a cell’s mechanical experience impacts its current and future shapes during a critical step of brain development.”
Beyond neural tube development, the findings highlight the power of physics-based modeling to explain complex biological processes that can’t be observed directly. The researchers say this approach could be applied to other stages of human development where forces, motion, and timing are just as critical.
Funding: The computational research at Banerjee Lab is funded by the National Institute of General Medical Sciences
Key Questions Answered:
A: Biology is governed by the laws of physics. Growing an organ requires moving matter, generating force, and managing tension. By treating the embryo as a mechanical system, physicists can measure the “load” and “stress” on cells, explaining how they move in ways a microscope alone cannot.
A: Yes. By identifying the specific mechanical “sweet spot” for closure, scientists can now look for environmental or chemical factors that might weaken the actin “drawstring” or disrupt the cellular synchronization, leading to better preventative strategies.
A: Highly likely. The researchers believe this type of force-driven coordination is a universal rule in development. Similar mechanisms are seen in how the body closes a wound or how certain organs, like the heart, take their final shape.
Editorial Notes:
- This article was edited by a Neuroscience News editor.
- Journal paper reviewed in full.
- Additional context added by our staff.
About this neuroscience research news
Author: Tess Malone
Source: Georgia Tech
Contact: Tess Malone – Georgia Tech
Image: The image is credited to Georgia Tech
Original Research: Open access.
“Mechanosensitive feedback organizes cell shape and motion during hindbrain neuropore morphogenesis” by Fernanda Pérez-Verdugo, Eirini Maniou, Gabriel L. Galea, and Shiladitya Banerjee. Current Biology
DOI:10.1016/j.cub.2026.02.068
Abstract
Mechanosensitive feedback organizes cell shape and motion during hindbrain neuropore morphogenesis
Neural tube closure is a critical morphogenetic process in vertebrate development, and failure to close cranial regions such as the hindbrain neuropore (HNP) leads to severe congenital malformations.
While mechanical forces such as actomyosin purse-string contraction and directional cell crawling have been implicated in driving HNP closure, how these forces organize local cell shape and motion to produce large-scale tissue remodeling remains poorly understood.
Using live and fixed imaging of mouse embryos combined with cell-based biophysical modeling, we show that these force-generating mechanisms are insufficient to explain the reproducible patterns of cell elongation and nematic alignment observed at the HNP border.
Instead, we show that local anisotropic stress and cytoskeletal organization are required to generate these patterns and promote midline cell motion.
Our model captures key features of cell shape dynamics and emergent nematic order, which we confirm experimentally, including the alignment of actin fibers with cell shape and enhanced midline cell speed.
Comparative analysis with chick embryos, which lack supracellular purse strings, supports a conserved link between tension generation and cellular patterning.
These findings establish a physical framework connecting force generation, cell shape anisotropy, and tissue morphodynamics during epithelial gap closure.