Summary: Study suggests an alternative strategy for restoring cellular abnormalities in in-vitro models of Parkinson’s disease.
Source: University of Luxembourg
In a seven-year research effort, an international team of scientists has clarified the cause for certain genetic forms of Parkinson’s disease, and has identified potential pharmacological treatments. The interdisciplinary research team, led by Prof. Rejko Krüger, of the Luxembourg Centre for Systems Biomedicine (LCSB) of the University of Luxembourg, experimented on patient-based cell cultures in the laboratory. The new combination of active substances they identified will have to undergo clinical trials before they can be used to treat patients.
The research team published its results today in the prestigious scientific journal Science Translational Medicine.
Lack of protein DJ-1 makes you sick
A protein called DJ-1 plays a crucial role in keeping nerve cells functioning. If the body is unable to produce ample amounts of DJ-1, important nerve cells die. The result is the onset of neuro-degenerative diseases such as Parkinson’s. The production of important proteins like DJ-1 can be disrupted or halted permanently if the genetic blueprints or the production processes they encode are defective.
Now, Prof. Rejko Krüger’s research team in Luxembourg has succeeded in identifying for the first time the importance of an error in the production process known as ‘splicing’ in the development of a certain form of Parkinson’s disease.
“In the patients, an essential tool for the assembly of the protein DJ-1 fails to dock properly,” Krüger explains.
“In scientific terms, we call that exon skipping. As a result of this defect, the protein doesn’t get built at all.”
The research result offers an entirely new point of attack for treating this malfunction of protein synthesis with drugs.
“This insight fundamentally changes our view of the causes of the disease and presents entirely new possibilities for treatment,” says Dr Ibrahim Boussaad, LCSB scientist and first author of the scientific paper.
“We could only gain this new understanding thanks to the skin cells from the patients,” Boussaad emphasises.
Cell donation enables progress
The Luxembourg Parkinson’s Study, initiated in 2015, includes a group of 800 Parkinson’s patients and 800 healthy control subjects.
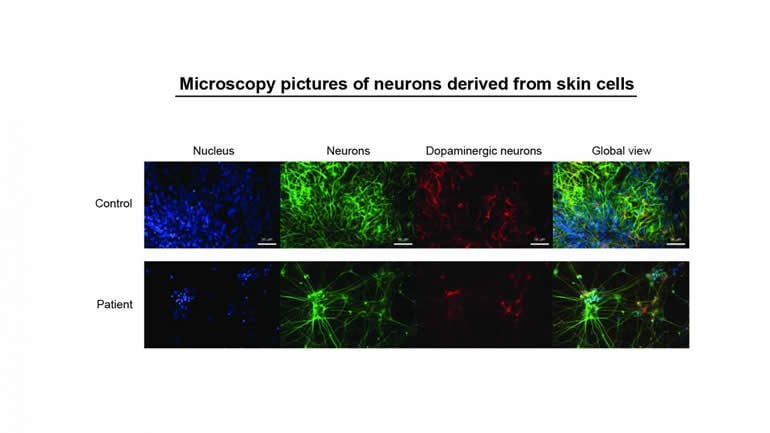
Thanks to the donation of skin cells taken by small biopsies, the researchers in Luxembourg were able to reprogram these cells to grow into nerve cells in vitro. These nerve cells are very similar to the neurons in affected regions of the donor’s brain and can be used for analyses and tests in the laboratory. Because it is not possible to take neurons directly from the brain of patients, for health and ethical reasons, reprogramming is the only way to examine the clinical features of the patient’s neurons in vitro. In scientific jargon, this is called a patient-based in vitro model, and is an important step in personalised medicine.
Using this method, Prof. Krüger’s team was able to explain the cause of the genetic form of Parkinson’s disease in which the PARK7 gene is mutated. Prof. Thomas Gasser, a medical director at the Tübingen University Hospital and co-author of the paper, adds, “We are proud to have been able to contribute our expertise in the reprogramming of patient cells to this stem cell work of our colleagues in Luxembourg.” Institutions from Germany, Italy and the USA collaborated in the research project.
Luxembourg’s interdisciplinarity is a key to this success
Precise bioinformatics algorithms developed at the LCSB allowed the research team to immediately carry out an automated search for potential active substances for drug treatment. This yielded a hit in the form of the active compounds phenylbutyric acid and RECTAS (RECTifier of Aberrant Splicing). Administered in combination, these two active substances allow the cells in the test tube to effectively reactivate the production of the important protein DJ-1.
“Only by combining numerous disciplines – from medical practice, to laboratory research, to computer science – could we understand the cause and at the same time identify active substances for a potential treatment,” Prof. Rejko Krüger explains.
He adds, “This kind of scientific progress ‘Made in Luxembourg’ is possible because all the necessary disciplines have been unified in Luxembourg for several years now.”
Funding: This work represents the high point to date of the PEARL program of the Luxembourg National Research Fund (FNR), through which the research of Prof. Krüger and his team is funded.
The team of scientists especially expresses its gratitude to the people who are participating in the Luxembourg Parkinson’s Study and who have made this research possible in the first place.
About this genetics research article
Source:
University of Luxembourg
Contacts:
Leslie Capo – University of Luxembourg
Image Source:
The image is credited to University of Luxembourg.
Original Research: Closed access
“A patient-based model of RNA mis-splicing uncovers treatment targets in Parkinson’s disease” by Boussaad et al. Science Translational Medicine.
Abstract
A patient-based model of RNA mis-splicing uncovers treatment targets in Parkinson’s disease
Parkinson’s disease (PD) is a heterogeneous neurodegenerative disorder with monogenic forms representing prototypes of the underlying molecular pathology and reproducing to variable degrees the sporadic forms of the disease. Using a patient-based in vitro model of PARK7-linked PD, we identified a U1-dependent splicing defect causing a drastic reduction in DJ-1 protein and, consequently, mitochondrial dysfunction. Targeting defective exon skipping with genetically engineered U1-snRNA recovered DJ-1 protein expression in neuronal precursor cells and differentiated neurons. After prioritization of candidate drugs, we identified and validated a combinatorial treatment with the small-molecule compounds rectifier of aberrant splicing (RECTAS) and phenylbutyric acid, which restored DJ-1 protein and mitochondrial dysfunction in patient-derived fibroblasts as well as dopaminergic neuronal cell loss in mutant midbrain organoids. Our analysis of a large number of exomes revealed that U1 splice-site mutations were enriched in sporadic PD patients. Therefore, our study suggests an alternative strategy to restore cellular abnormalities in in vitro models of PD and provides a proof of concept for neuroprotection based on precision medicine strategies in PD.







