Summary: Researchers identify a group of neurons that are activated by oxytocin in an area of the female brain, but which are not present in the same area of a male mouse brain. Altered expression of these neurons may result in postpartum depression.
Source: LSU
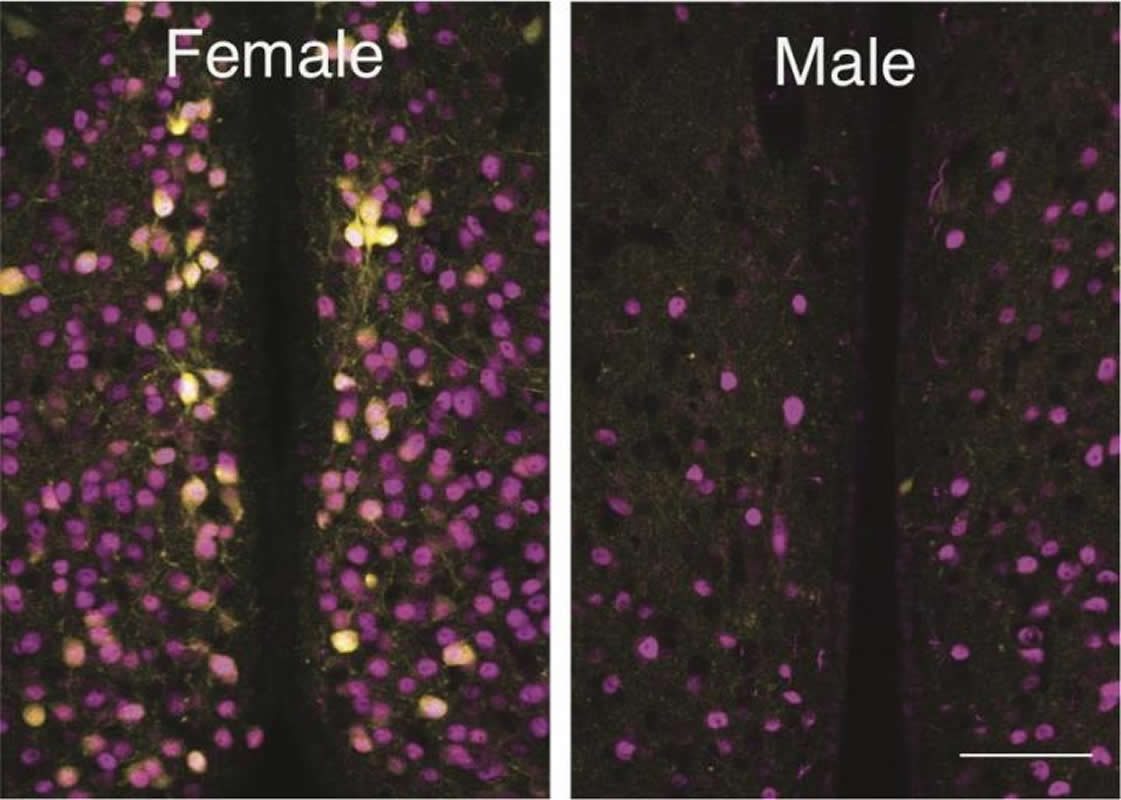
Oxytocin is widely referred to as the love hormone and plays an important role in the regulation of social and maternal behavior. In recent years, the oxytocin system in the brain has received tremendous attention as key to new treatments for many mental health disorders, such as anxiety, autism spectrum disorders and postpartum depression. New research led by a biologist and his students at LSU have discovered a group of cells that are activated by oxytocin in one area of female mouse brains that are not present in the same area in male mouse brains.
“Many researchers have attempted to investigate the difference between the oxytocin system in females versus males, but no one has successfully found conclusive evidence until now. Our discovery was a big surprise,” said Ryoichi Teruyama, LSU Department of Biological Sciences associate professor, who led this study published in PLOS ONE.
The oxytocin receptor cells are present in the brain area thought to be involved in the regulation of maternal behavior. Moreover, the expression of oxytocin receptors in these cells are only present when estrogen is also present. These imply that these cells are involved in inducing maternal behavior. In addition, it confirms what many recent human studies have shown: there is a connection between altered expression of oxytocin receptors and postpartum depression.
Postpartum depression contributes to poor maternal health and has negative effects on a child’s development. A number of studies have found that children of depressed mothers are at risk for a wide range of cognitive, emotional, behavioral and medical problems. Therefore, postpartum depression is a major public health concern that has significant adverse effects on both mother and child. About 10 to 20 percent of women experience postpartum depression after childbirth.
This new discovery that occurred at LSU opens doors to potential new treatments and drugs for postpartum depression targeting oxytocin receptor cells.
“I think our discovery could be universal to all mammals that exhibit maternal behavior, including humans,” Teruyama said.
Student researchers
Study co-author Ryan LeBlanc from Denham Springs was an undergraduate student researcher at LSU whose work was instrumental to this discovery. However, he had little previous research experience before joining Teruyama’s lab.
Teruyama recalled that when LeBlanc first approached him to be his mentor, he asked him about his hobbies. LeBlanc said he liked to build plastic models of battleships.
“I certainly don’t know much about battleship plastic models, but anyone who can assemble 500 to 2,000 plastic parts into models must be persistent, focused and exceedingly careful. I accepted him gladly thinking he is going to find something extraordinary, and I was right,” Teruyama said.
LeBlanc took on the tedious task of finding and marking the exact location of thousands of oxytocin receptor cells with a red pen. He spent more than a month identifying the cells, which was instrumental to this discovery.
Current LSU doctoral candidate Kaustubh Sharma from Nepal is the first author who validated LeBlanc’s finding. Sharma is currently investigating if the oxytocin receptor cells regulate maternal behavior in female mice.
Source:
LSU
Media Contacts:
Alison Satake – LSU
Image Source:
The image is credited to Ryoichi Teruyama, LSU.
Original Research: Open access
“Sexually dimorphic oxytocin receptor-expressing neurons in the preoptic area of the mouse brain”. Kaustubh Sharma, Ryan LeBlanc, Masudul Haque, Katsuhiko Nishimori, Madigan M. Reid, Ryoichi Teruyama.
PLOS ONE. doi:10.1371/journal.pone.0219784
Abstract
Sexually dimorphic oxytocin receptor-expressing neurons in the preoptic area of the mouse brain
Oxytocin is involved in the regulation of social behaviors including parental behaviors in a variety of species. Oxytocin triggers social behaviors by binding to oxytocin receptors (OXTRs) in various parts of the brain. OXTRs are present in the preoptic area (POA) where hormone-sensitive sexually dimorphic nuclei exist. The present study was conducted to examine whether sex differences exist in the distribution of neurons expressing OXTRs in the POA. Using OXTR-Venus (an enhanced variant of yellow fluorescent protein) mice, the distribution of OXTR-Venus cells in the POA was compared between sexes. The total number of OXTR-Venus cells in the medial POA (MPOA) was significantly greater in females than in males. No detectable OXTR-Venus cells were observed in the anteroventral periventricular nucleus (AVPV) within the MPOA in most of the brain sections from males. We further examined the total number of OXTR-Venus cells in the AVPV and the rest of the MPOA between the sexes. The total number of OXTR-Venus cells in the AVPV in females (615 ± 43) was significantly greater than that in males (14 ± 2), whereas the total number of OXTR-Venus cells in the rest of the MPOA did not differ significantly between the sexes. Thus, the sexually dimorphic expression of OXTR-Venus specifically occurred in the AVPV, but not in the rest of the MPOA. We also examined whether the expression of OXTR in the AVPV is driven by the female gonadal hormone, estrogen. Immunocytochemistry and single-cell RT-PCR revealed the presence of the estrogen receptor α in OXTR-Venus cells in the female AVPV. Moreover, ovariectomy resulted in the absence of OXTR-Venus expression in the AVPV, whereas estrogen replacement therapy restored OXTR-Venus expression. These results demonstrate that the expression of OXTR in the AVPV is primarily female specific and estrogen dependent. The presence of the sexually dimorphic expression of OXTR in the AVPV suggests the involvement of OXTR neurons in the AVPV in the regulation of female-specific behavior and/or physiology.






