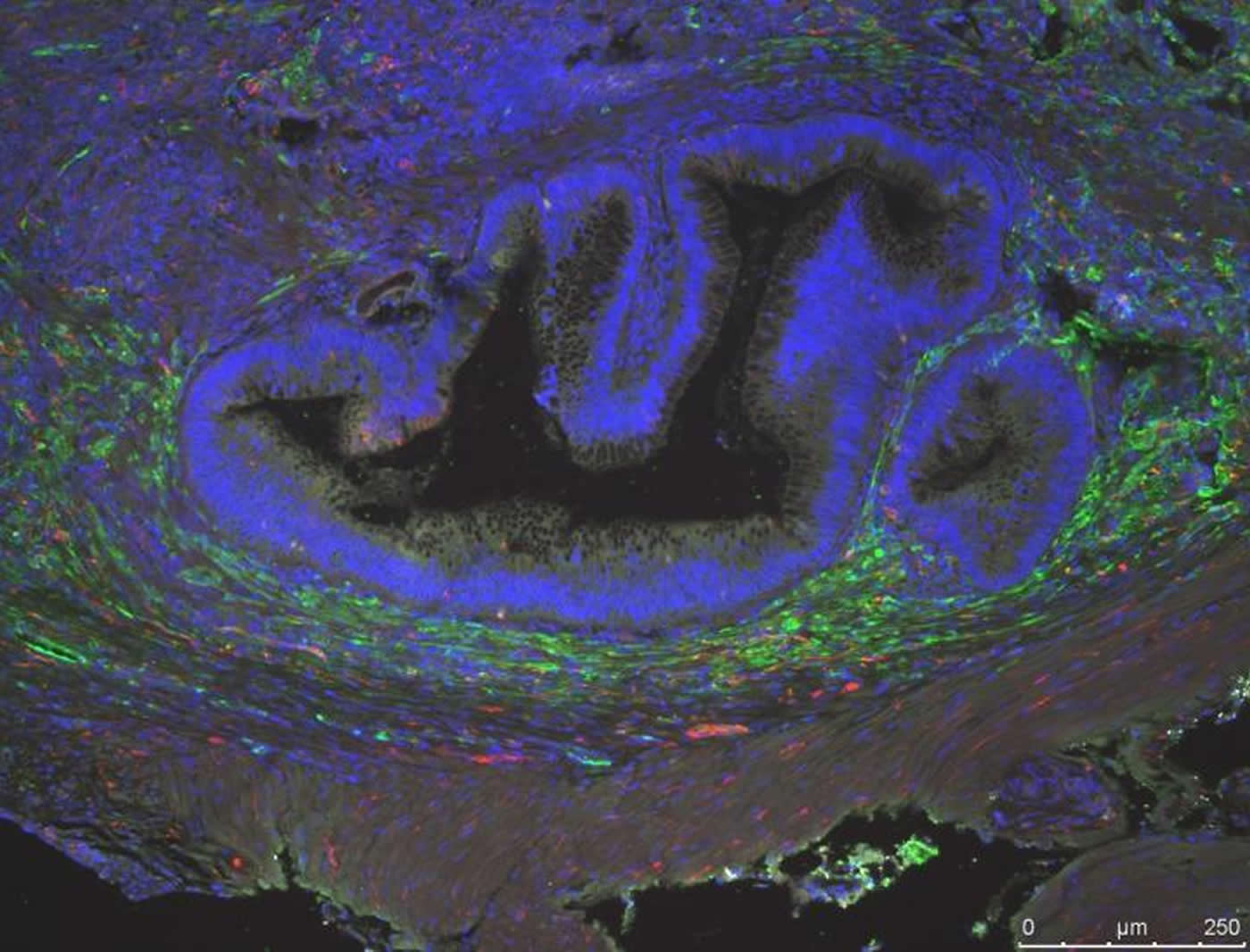
A study by scientists at Children’s Hospital Los Angeles has shown that tissue-engineered colon derived from human cells is able to develop the many specialized nerves required for function, mimicking the neuronal population found in native colon. These specialized neurons, localized in the gut, form the enteric nervous system, which regulates digestive tract motility, secretion, absorption and gastrointestinal blood flow. In addition, in a condition called Hirschsprung’s disease or aganglionosis, where those neurons are not present, the team was able to replace them.
The study — the first report on the enteric nervous system in human-derived tissue-engineered colon — was published online ahead of print in the journal of Tissue Engineering, Part A on September 28.
In healthy intestines, food is moved along the digestive tract through peristalsis — a series of wave-like contractions. Special nerve cells called ganglion cells are required for this movement, but there is also a rich mixture of other types of nerve cells. In children with Hirschsprung’s disease, these cells are missing. Without them, the intestine becomes blocked and surgical removal of the affected segment of colon is required.
To help these and other children suffering from intestinal diseases that may require surgical removal of all or part of their intestine, the CHLA team — led by principal investigator Tracy C. Grikscheit, MD, a pediatric surgeon and researcher at The Saban Research Institute of CHLA — is developing tissue-engineered options for these children.
One objective of growing tissue-engineered organs is to generate new tissue from a patient’s own cells. Grikscheit and her team first needed to determine what parts of the enteric nervous system were present in tissue-engineered colon when it is grown from normal human cells.
“The diversity of neuron types that grew within the human tissue-engineered colon was a revelation to our team, because previously we had only documented that some ganglia were present,” said Grikscheit, who is also a tenured associate professor of Surgery at the Keck School of Medicine of the University of Southern California. “The next step was to determine if these neuronal elements could be supplied to tissue-engineered colon that was missing neurons – like in Hirschsprung’s disease.”

The scientists initially grew cells from patients with Hirschsprung’s disease and from mice with a genetic mutation that causes aganglionosis. In both cases, the tissue-engineered colon derived from these cells did not have the all-important components of the intestinal nervous system. In a second set of experiments, again testing both mouse and human cells, the investigators added neurospheres, which are clusters of purified neural progenitor cells. The cells had been stained with green fluorescence, so the scientists could readily visualize where the nerve cells ended up in the tissue-engineered colon, as well as determine the source of the nerve cells.
“After growing the colon for four weeks, we saw that the green nerve cells had been incorporated into the colon engineered from human tissue derived from a patient lacking those elements and that the different nerve subtypes were present,” said first author on the study, Minna Wieck, MD, an investigator and surgical resident at CHLA.
Additional contributors to the study include Wael N. El-Nachef, Xiaogang Hou, Ryan G. Spurrier, Kathleen A. Holoyda, Kathy A. Schall, Salvador Garcia Mojica, Malie K. Collins, and Andrew Trecartin of Children’s Hospital Los Angeles; and Zhi Cheng, and Philip K. Frykman of Cedars-Sinai Medical Center, Los Angeles.
Funding: Funding was provided in part by the California Institute of Regenerative Medicine (RN3 00946-1, RN3 06425, TG2-01168) and from the National Institute of Diabetes, Digestive and Kidney Diseases (K08DK090281).
Source: Ellin Kavanagh – Children’s Hospital of Los Angeles
Image Source: The image is credited to Children’s Hospital of Los Angeles
Original Research: Abstract for “Human and murine tissue-engineered colon exhibit diverse neuronal subtypes and can be populated by enteric nervous system progenitor cells when donor colon is aganglionic” by Dr. Minna M Wieck, Dr. Wael N El-Nachef, Dr. Xiaogang Hou, Dr. Ryan Gregory Spurrier, Dr. Kathleen A Holoyda, Dr. Kathy A Schall, Mr. Salvador Mojica Garcia, Miss Malie K Collins, Dr. Andrew Trecartin, Dr. Zhi Cheng, Dr. Philip K Frykman, and Dr. Tracy C Grikscheit in Tissue Engineering Part A. Published online September 28 2015 doi:10.1089/ten.TEA.2015.0120
Abstract
Human and murine tissue-engineered colon exhibit diverse neuronal subtypes and can be populated by enteric nervous system progenitor cells when donor colon is aganglionic
Neurons live for decades in a postmitotic state, their genomes susceptible to DNA damage. Here we survey the landscape of somatic single-nucleotide variants (SNVs) in the human brain. We identified thousands of somatic SNVs by single-cell sequencing of 36 neurons from the cerebral cortex of three normal individuals. Unlike germline and cancer SNVs, which are often caused by errors in DNA replication, neuronal mutations appear to reflect damage during active transcription. Somatic mutations create nested lineage trees, allowing them to be dated relative to developmental landmarks and revealing a polyclonal architecture of the human cerebral cortex. Thus, somatic mutations in the brain represent a durable and ongoing record of neuronal life history, from development through postmitotic function.
“Human and murine tissue-engineered colon exhibit diverse neuronal subtypes and can be populated by enteric nervous system progenitor cells when donor colon is aganglionic” by Dr. Minna M Wieck, Dr. Wael N El-Nachef, Dr. Xiaogang Hou, Dr. Ryan Gregory Spurrier, Dr. Kathleen A Holoyda, Dr. Kathy A Schall, Mr. Salvador Mojica Garcia, Miss Malie K Collins, Dr. Andrew Trecartin, Dr. Zhi Cheng, Dr. Philip K Frykman, and Dr. Tracy C Grikscheit in Tissue Engineering Part A. Published online September 28 2015 doi:10.1089/ten.TEA.2015.0120