Summary: Following a meal, astrocytes associated with POMC neurons in the hypothalamus alter their shape. After eating, glucose levels increase temporarily. Astrocytes detect the signal and react within one hour, causing POMC neurons to activate and promote the feeling of satiety.
Source: CNRS
You just finished a good meal and are feeling full? Researchers from the CNRS, Inrae, University of Burgundy, Université de Paris, Inserm, and the University of Luxembourg have just revealed the mechanisms in our brains that lead to this state. They involve a series of reactions triggered by a rise in blood glucose levels. This study, which was conducted on mice, is published in Cell Reports on 3 March 2020.
The neuronal circuits in our brain governing feelings of hunger and satiety can modify their connections, thereby adjusting feeding behavior to living conditions and maintaining a balance between food intake and calorie expenditure. Scientists suspect that this plasticity could be altered for obese subjects.
In a new study conducted on mice, a team led by Alexandre Benani, a CNRS researcher at the Centre for Taste and Feeding Behavior (CNRS/Inrae/University of Burgundy/AgroSup Dijon), has shown that these circuits are activated on the time scale of a meal, subsequently regulating feeding behavior. However, this activation does not occur through a change in the circuit’s “connections.”
Scientists focused on POMC neurons in the hypothalamus, located at the base of the brain, which is known for limiting food intake. They are connected to a large number of neurons from other parts of the brain, with the connections of this circuit being malleable: they can be made and unmade very quickly based on hormonal fluctuations. Researchers observed that this neuronal circuit is not modified after a balanced meal, but that other cells associated with POMC neurons, known as astrocytes, actually change form.
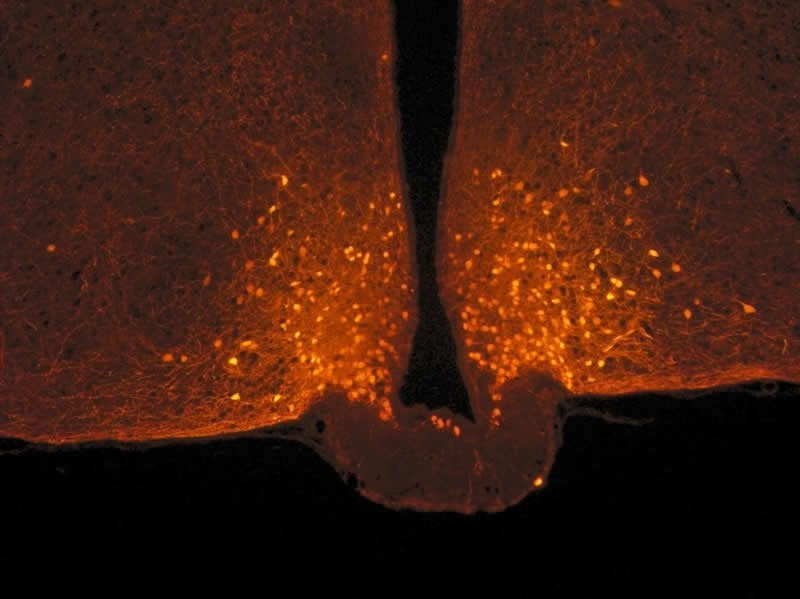
Astrocytes are star-shaped cells that were first studied for their supporting role with respect to neurons. Under usual conditions, they sheathe POMC neurons and act somewhat like brake pads by limiting their activity. After a meal, blood glucose levels (glycemia) temporarily increase, with astrocytes detecting this signal and retracting in less than one hour: once this “brake” is released, POMC neurons are activated, ultimately promoting the feeling of satiety.
Surprisingly, a meal that is high in fats does not lead to this remodeling. Does this mean that lipids are less effective in satisfying hunger? The scientists are trying to determine whether they trigger satiety through another circuit. It also remains to be seen whether sweeteners have the same effects, or whether they lure the brain by providing an addictive sensation of sweetness without satisfying hunger.
Source:
CNRS
Media Contacts:
Veronique Etienne – CNRS
Image Source:
The image is credited to Danaé Nuzzaci / CNRS / CSGA.
Original Research: Open access
“Postprandial Hyperglycemia Stimulates Neuroglial Plasticity in Hypothalamic POMC Neurons after a Balanced Meal”. Danaé Nuzzaci, Céline Cansell, Fabienne Liénard, Emmanuelle Nédélec, Selma Ben Fradj, Julien Castel, Ewout Foppen, Raphael Denis, and others.
Cell Reports doi:10.1016/j.celrep.2020.02.029.
Abstract
Postprandial Hyperglycemia Stimulates Neuroglial Plasticity in Hypothalamic POMC Neurons after a Balanced Meal
Highlights
• Postprandial hyperactivity of hypothalamic POMC neurons involves synaptic plasticity
• Postprandial plasticity of POMC neurons engages glial retraction
• Postprandial glial retraction around POMC neurons is triggered by hyperglycemia
• Glial retraction on POMC neurons modifies meal pattern
Summary
Mechanistic studies in rodents evidenced synaptic remodeling in neuronal circuits that control food intake. However, the physiological relevance of this process is not well defined. Here, we show that the firing activity of anorexigenic POMC neurons located in the hypothalamus is increased after a standard meal. Postprandial hyperactivity of POMC neurons relies on synaptic plasticity that engages pre-synaptic mechanisms, which does not involve structural remodeling of synapses but retraction of glial coverage. These functional and morphological neuroglial changes are triggered by postprandial hyperglycemia. Chemogenetically induced glial retraction on POMC neurons is sufficient to increase POMC activity and modify meal patterns. These findings indicate that synaptic plasticity within the melanocortin system happens at the timescale of meals and likely contributes to short-term control of food intake. Interestingly, these effects are lost with a high-fat meal, suggesting that neuroglial plasticity of POMC neurons is involved in the satietogenic properties of foods.






