Summary: Researchers use coherent control to control the function of a living cell.
Source: University of Illinois a Urbana Champaign.
Specially tailored, ultrafast pulses of light can trigger neurons to fire and could one day help patients with light-sensitive circadian or mood problems, according to a new study in mice at the University of Illinois.
Chemists have used such carefully crafted light beams, called coherent control, to regulate chemical reactions, but this study is the first demonstration of using them to control function in a living cell. The study used optogenetic mouse neurons – that is, cells that had a gene added to make them respond to light. However, the researchers say the same technique could be used on cells that are naturally responsive to light, such as those in the retina.
“The saying, ‘The eye is the window to the soul’ has some merit, because our bodies respond to light. Photoreceptors in our retinas connect to different parts in the brain that control mood, metabolic rhythms and circadian rhythms,” said Dr. Stephen Boppart, the leader of the study published in the journal Nature Physics. Boppart is an Illinois professor of electrical and computer engineering and of bioengineering, and also is a medical doctor.
The researchers used light to excite a light-sensitive channel in the membrane of neurons. When the channels were excited, they allowed ions through, which caused the neurons to fire.
While most biological systems in nature are accustomed to the continuous light from the sun, Boppart’s team used a flurry of very short light pulses – less than 100 femtoseconds. This delivers a lot of energy in a short period of time, exciting the molecules to different energy states. Along with controlling the length of the light pulses, Boppart’s team controls the order of wavelengths in each light pulse.
“When you have an ultrashort or ultrafast pulse of light, there’s many colors in that pulse. We can control which colors come first and how bright each color will be,” Boppart said. “For example, blue wavelengths are much higher energy than red wavelengths. If we choose which color comes first, we can control what energy the molecule sees at what time, to drive the excitement higher or back down to the base line. If we create a pulse where the red comes before the blue, it’s very different than if the blue comes before the red.”
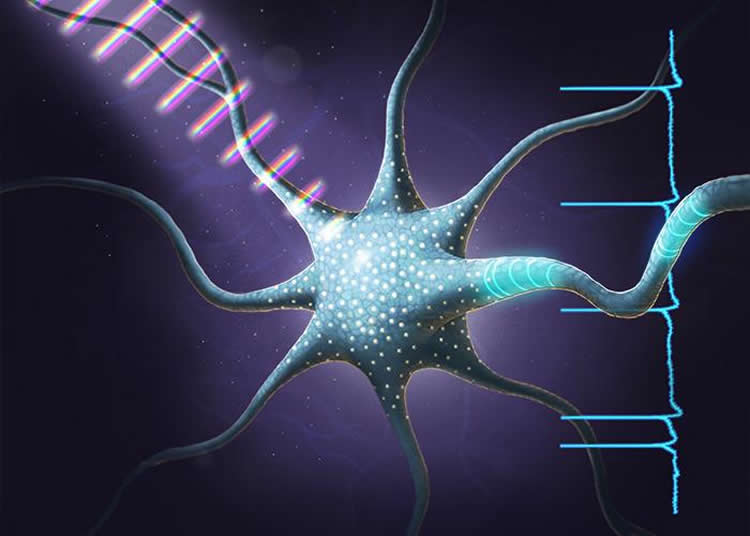
The researchers demonstrated using patterns of tailored light pulses to make the neurons fire in different patterns.
Boppart says coherent control could give optogenetics studies more flexibility, since changing properties of the light used can give researchers more avenues than having to engineer mice with new genes every time they want a different neuron behavior.
Outside of optogenetics, the researchers are working to test their coherent control technique with naturally light-responsive cells and processes – retinal cells and photosynthesis, for example.
“What we’re doing for the very first time is using light and coherent control to regulate biological function. This is fundamentally more universal than optogenetics – that’s just the first example we used,” Boppart said. “Ultimately, this could be a gene-free, drug-free way of regulating cell and tissue function. We think there could be ‘opto-ceuticals,’ methods of treating patients with light.”
Funding: Funding provided by National Science Foundation, National Institutes of Health.
Source: Liz Ahlberg Touchstone – University of Illinois a Urbana Champaign
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Stephen Boppart, University of Illinois.
Video Source:: Video credited to MMHI Howard Hughes Medical Institute.
Original Research: Full open access research for “Coherent control of an opsin in living brain tissue” by Kush Paul, Parijat Sengupta, Eugene D. Ark, Haohua Tu, Youbo Zhao & Stephen A. Boppart in Nature Physics. Published online September 18 2017 doi:10.1038/nphys4257
[cbtabs][cbtab title=”MLA”]University of Illinois a Urbana Champaign “Carefully Crafted Light Pulses Control Neuron Activity.” NeuroscienceNews. NeuroscienceNews, 17 November 2017.
<https://neurosciencenews.com/neuron-activity-light-7976/>.[/cbtab][cbtab title=”APA”]University of Illinois a Urbana Champaign (2017, November 17). Carefully Crafted Light Pulses Control Neuron Activity. NeuroscienceNews. Retrieved November 17, 2017 from https://neurosciencenews.com/neuron-activity-light-7976/[/cbtab][cbtab title=”Chicago”]University of Illinois a Urbana Champaign “Carefully Crafted Light Pulses Control Neuron Activity.” https://neurosciencenews.com/neuron-activity-light-7976/ (accessed November 17, 2017).[/cbtab][/cbtabs]
Abstract
Coherent control of an opsin in living brain tissue
Retinal-based opsins are light-sensitive proteins. The photoisomerization reaction of these proteins has been studied outside cellular environments using ultrashort tailored light pulses1,2,3,4,5. However, how living cell functions can be modulated via opsins by modifying fundamental nonlinear optical properties of light interacting with the retinal chromophore has remained largely unexplored. We report the use of chirped ultrashort near-infrared pulses to modulate light-evoked ionic current from Channelrhodopsin-2 (ChR2) in brain tissue, and consequently the firing pattern of neurons, by manipulating the phase of the spectral components of the light. These results confirm that quantum coherence of the retinal-based protein system, even in a living neuron, can influence its current output, and open up the possibilities of using designer-tailored pulses for controlling molecular dynamics of opsins in living tissue to selectively enhance or suppress neuronal function for adaptive feedback-loop applications in the future.
“Coherent control of an opsin in living brain tissue” by Kush Paul, Parijat Sengupta, Eugene D. Ark, Haohua Tu, Youbo Zhao & Stephen A. Boppart in Nature Physics. Published online September 18 2017 doi:10.1038/nphys4257






