Summary: A newly developed technique allows researchers to remotely active neurons with the aid of microscopic magnetic particles.
Source: UCL
Scientists at UCL have developed a new technique that uses microscopic magnetic particles to remotely activate brain cells; researchers say the discovery in rats could potentially lead to the development of a new class of non-invasive therapies for neurological disorders.
Published in Advanced Science, the pioneering technique called “magnetomechanical stimulation” or , allows touch sensitive brain glial cells called astrocytes to be stimulated with a magnetic device outside the body.
Microscopic magnetic particles, or micromagnets, are attached to astrocytes, and used as miniature mechanical switches that can turn “on” the cells when a strong magnet is placed near the head.
Co-author, Professor Alexander Gourine (UCL Centre for Cardiovascular and Metabolic Neuroscience) said: “Astrocytes are star-shaped cells found throughout the brain. They are strategically positioned between the brain blood vessels and nerve cells. These cells provide neurons with essential metabolic and structural support, modulate neuronal circuit activity and may also function as versatile surveyors of brain milieu, tuned to sense conditions of potential metabolic insufficiency.
“The ability to control brain astrocytes using a magnetic field gives the researchers a new tool to study the function of these cells in health and disease that may be important for future development of novel and effective treatments for some common neurological disorders, such as epilepsy and stroke.”
Senior author, Professor Mark Lythgoe (UCL Centre for Advanced Biomedical Imaging) said: “Because astrocytes are sensitive to touch, decorating them with magnetic particles means you can give the cells a tiny prod from outside the body using a magnet, and as such, control their function. This ability to remotely control astrocytes provides a new tool for understanding their function and may have the potential to treat brain disorders.”
In developing MMS, scientists at UCL set out to create a more clinically relevant brain cell control technique. This contrasts with other existing research tools, such as optogenetics and chemogenetics, which require foreign genes to be inserted into the brain cells, typically with the help of a virus. This need for genetic modification has been a major obstacle to the clinical translation of the existing methods.
Lead researcher Dr. Yichao Yu (UCL Centre for Advanced Biomedical Imaging) said: “Our new technology uses magnetic particles and magnets to remotely and precisely control brain cell activity and, importantly, does this without introducing any device or foreign gene into the brain.
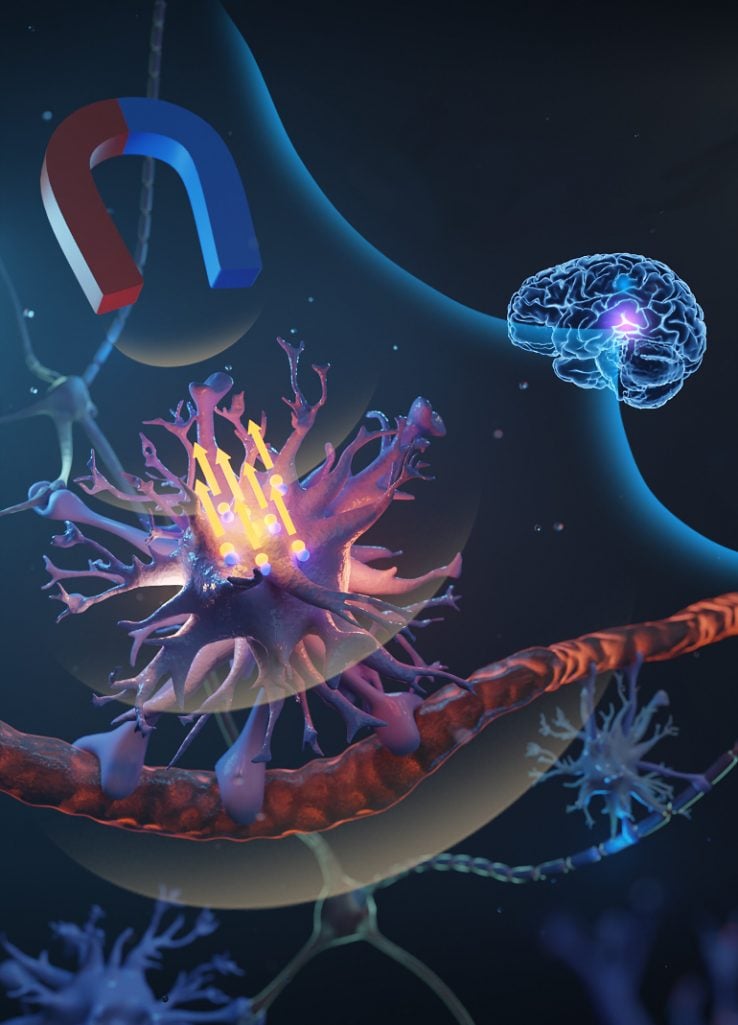
“In the laboratory-based study, we coated microscopic magnetic particles with an antibody that enables them to bind specifically to astrocytes. The particles were then delivered to the target brain region in the rat via injection.
“Another advantage of using micromagnets is that they light up on an MRI scan so we can track their location and target very particular parts of the brain to get precise control of brain function.”
Professor Lythgoe, who received the Royal Society of Medicine Ellison–Cliffe Award 2021 for his “contribution of fundamental science to the advancement of medicine”, added:
“We are very excited about this technology because of its clinical potential. In contrast to existing methods, MMS takes advantage of the remarkable sensitivity to touch of certain brain cells, therefore neither genetic modification nor device implantation is needed. This makes MMS a promising candidate as an alternative, less invasive therapy compared to the currently used deep brain stimulation techniques that require the insertion of electrodes into the brain.”
About this neurotech research news
Author: Press Office
Source: UCL
Contact: Press Office – UCL
Image: The image is credited to Yichao Yu and Mark Lythgoe at UCL
Original Research: Open access.
“Remote and Selective Control of Astrocytes by Magnetomechanical Stimulation” by Yichao Yu et al. Advanced Science
Abstract
Remote and Selective Control of Astrocytes by Magnetomechanical Stimulation
Astrocytes play crucial and diverse roles in brain health and disease. The ability to selectively control astrocytes provides a valuable tool for understanding their function and has the therapeutic potential to correct dysfunction. Existing technologies such as optogenetics and chemogenetics require the introduction of foreign proteins, which adds a layer of complication and hinders their clinical translation.
A novel technique, magnetomechanical stimulation (MMS), that enables remote and selective control of astrocytes without genetic modification is described here. MMS exploits the mechanosensitivity of astrocytes and triggers mechanogated Ca2+ and adenosine triphosphate (ATP) signaling by applying a magnetic field to antibody-functionalized magnetic particles that are targeted to astrocytes.
Using purpose-built magnetic devices, the mechanosensory threshold of astrocytes is determined, a sub-micrometer particle for effective MMS is identified, the in vivo fate of the particles is established, and cardiovascular responses are induced in rats after particles are delivered to specific brainstem astrocytes.
By eliminating the need for device implantation and genetic modification, MMS is a method for controlling astroglial activity with an improved prospect for clinical application than existing technologies.






