Summary: Chronic exposure to manganese, as experienced by some working as industrial welders, has been linked to the onset of Parkinson’s like symptoms. Researchers report manganese exposure can cause misfolded alpha-synuclein, a hallmark of Parkinson’s disease.
Source: Iowa State University
A new study from Iowa State University biomedical researchers illuminates the biological processes by which exposure to some metals can contribute to the onset of Parkinson’s-like symptoms.
The study, published today in the peer-reviewed journal Science Signaling, focuses on the metal manganese, which has a range of industrial uses as an alloy. Anumantha Kanthasamy, a Clarence Hartley Covault Distinguished Professor in veterinary medicine and the Eugene and Linda Lloyd Endowed Chair of Neurotoxicology, said the research details how manganese exposure can lead to misfolded proteins in the brain, which cause a neurological disease. Kanthasamy said the findings could lead to earlier detection of the disease and better outcomes for patients.
Kanthasamy said small amounts of manganese are necessary for the proper functioning of the human body, but too much exposure has been linked with neurological symptoms much like those experienced by patients with Parkinson’s Disease. Links between manganese and neurological disorders have been noted since the 1950s, Kanthasamy said, because of the tendency of manganese to accumulate in brain tissues.
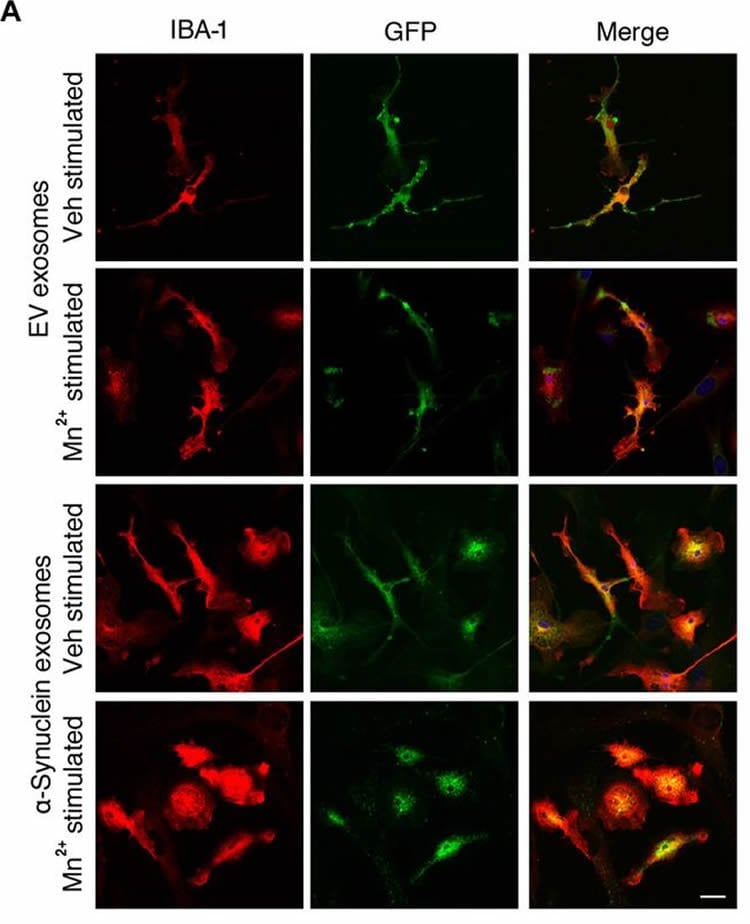
The latest study found that manganese combines with a protein in the brain called alpha-synuclein. Previous studies showed the protein was susceptible to misfolding, but Kanthasamy and his colleagues set out to discover how it interacted with manganese and how that interaction facilitates the progression of the disease. The researchers found the pathological form of misfolded alpha-synuclein proteins get packaged into vesicles, which allow the misfolded proteins to transfer from cell to cell to propagate the protein-seeding activity. These vesicles provoke inflammation of tissues and can lead to a neurodegenerative response, the study found.
The study drew on data gathered from mice as well as blood serum samples from welders provided by clinicians at Penn State University. The study found welders exposed to manganese had increased misfolded alpha-synuclein serum content, meaning the welders are at a higher risk for developing Parkinson’s symptoms, Kanthasamy said.
The research could contribute to a new assay, or medical test, to detect the presence of misfolded alpha-synuclein proteins. This could lead to earlier detection of Parkinson’s Disease and a way to gauge the effectiveness of drugs designed to slow the disease.
“As the disease advances, it’s harder to slow it down with treatments,” Kanthasamy said. “Earlier detection, perhaps by testing for misfolded alpha-synuclein, can lead to better outcomes for patients. Such a test might also indicate whether someone is at risk before the onset of the disease.”
Kanthasamy cautioned the research is still at an experimental stage, meaning it could be years before such an assay could be available.
Dilshan S. Harischandra, a former member of Kanthasamy’s lab who now works at the University of Pennsylvania, was the lead author of the study. Kanthasmy, chair of the Department of Biomedical Sciences in the ISU College of Veterinary Medicine, was the senior author of the study.
Funding: The study was supported by a grant from the National Institute of Environmental Health Sciences.
Source:
Iowa State University
Media Contacts:
Fred Love – Iowa State University
Image Source:
The image is credited to Kanthasamy et al./Science Signaling.
Original Research:
“Manganese promotes the aggregation and prion-like cell-to-cell exosomal transmission of α-synuclein”
BY DILSHAN S. HARISCHANDRA, DHARMIN ROKAD, MATTHEW L. NEAL, SHIVANI GHAISAS, SIREESHA MANNE, SOUVARISH SARKAR, NIKHIL PANICKER, GARY ZENITSKY, HUAJUN JIN, MECHELLE LEWIS, XUEMEI HUANG, VELLAREDDY ANANTHARAM, ARTHI KANTHASAMY, ANUMANTHA G. KANTHASAMY
Science Signaling.12 MAR 2019 doi:10.1126/scisignal.aau4543
Abstract
Manganese promotes the aggregation and prion-like cell-to-cell exosomal transmission of α-synuclein
The aggregation of α-synuclein (αSyn) is considered a key pathophysiological feature of certain neurodegenerative disorders, collectively termed synucleinopathies. Given that a prion-like, cell-to-cell transfer of misfolded αSyn has been recognized in the spreading of αSyn pathology in synucleinopathies, we investigated the biological mechanisms underlying the propagation of the disease with respect to environmental neurotoxic stress. Considering the potential role of the divalent metal manganese (Mn2+) in protein aggregation, we characterized its effect on αSyn misfolding and transmission in experimental models of Parkinson’s disease. In cultured dopaminergic neuronal cells stably expressing wild-type human αSyn, misfolded αSyn was secreted through exosomes into the extracellular medium upon Mn2+ exposure. These exosomes were endocytosed through caveolae into primary microglial cells, thereby mounting neuroinflammatory responses. Furthermore, Mn2+-elicited exosomes exerted a neurotoxic effect in a human dopaminergic neuronal model (LUHMES cells). Moreover, bimolecular fluorescence complementation (BiFC) analysis revealed that Mn2+ accelerated the cell-to-cell transmission of αSyn, resulting in dopaminergic neurotoxicity in a mouse model of Mn2+ exposure. Welders exposed to Mn2+ had increased misfolded αSyn content in their serum exosomes. Stereotaxically delivering αSyn-containing exosomes, isolated from Mn2+-treated αSyn-expressing cells, into the striatum initiated Parkinsonian-like pathological features in mice. Together, these results indicate that Mn2+ exposure promotes αSyn secretion in exosomal vesicles, which subsequently evokes proinflammatory and neurodegenerative responses in both cell culture and animal models.






