Activating small group of neurons in medulla causes rapid transition to REM sleep.
At the flip of a switch, University of California, Berkeley, neuroscientists can send a sleeping mouse into dreamland.
The researchers inserted an optogenetic switch into a group of nerve cells located in the ancient part of the brain called the medulla, allowing them to activate or inactivate the neurons with laser light.
When the neurons were activated, sleeping mice entered REM sleep within seconds. REM sleep, characterized by rapid eye movements, is the dream state in mammals accompanied by activation of the cortex and total paralysis of the skeletal muscles, presumably so that we don’t act out the dreams flashing through our mind.
Inactivating the neurons reduced or even eliminated a mouse’s ability to enter REM sleep.
“People used to think that this region of the medulla was only involved in the paralysis of skeletal muscles during REM sleep,” said lead author Yang Dan, a UC Berkeley professor of molecular and cell biology and a Howard Hughes Medical Institute Investigator. “What we showed is that these neurons triggered all aspects of REM sleep, including muscle paralysis and the typical cortical activation that makes the brain look more awake than in non-REM sleep.”
While other types of neurons in the brainstem and hypothalamus have been shown to influence REM sleep, Dan said, “Because of the strong induction of REM sleep – in 94 percent of the recorded trials our mice entered REM sleep within seconds of activating the neurons – we think this might be a critical node of a relatively small network that makes the decision whether you go into dream sleep or not.”
The UC Berkeley team reported their results in the Oct. 15 print issue of the British journal Nature, and the paper was posted online Oct. 7.
The discovery will not only help researchers better understand the complex control of sleep and dreaming in the brain, the researchers said, but will allow scientists to stop and start dreaming at will in mice to learn why we dream.
“Many psychiatric disorders, especially mood disorders, are correlated with changes in REM sleep, and some widely used drugs affect REM sleep, so it seems to be a sensitive indicator of mental and emotional health,” said first author Franz Weber, a UC Berkeley postdoctoral fellow. “We are hoping that studying the sleep circuit might lead us to new insights into these disorders as well as neurological diseases that affect sleep, like Parkinson’s and Alzheimer’s diseases.”
Eating and dreaming
The researchers also found that activating these brain cells while the mice were awake had no effect on wakefulness, but did make them eat more. In normal mice, these neurons – a subset of nerve cells that release the neurotransmitter gamma-amino butyric acid (GABA), and so are called GABAergic neurons – are most active during waking periods when the mice are eating or grooming, two highly pleasurable activities.
Dan suspects that these GABAergic neurons in the medulla have the opposite effect of stress neurons, such as the noradrenergic neurons in the pons, another ancient part of the brain. Noradrenergic neurons release the transmitter noradrenalin, a cousin of adrenalin.
“Other people have found that noradrenergic neurons, which are active when you are running, shut down when eating or grooming. So it seems like when you are relaxed and enjoying yourself, the noradrenergic neurons switch off and these GABAergic neurons in the medulla turn on,” she said.
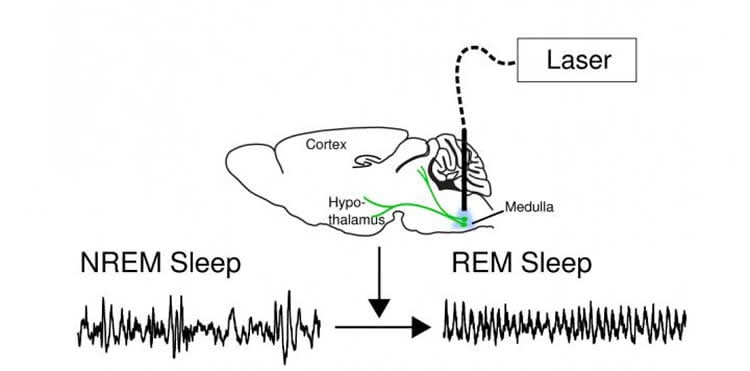
The GABAergic neurons project from the ventral part of the medulla, which sits at the top of the spinal cord, into many regions of the brainstem and hypothalamus, and thus are able to affect many bodily functions. These regions – more primitive than the brain’s cortex, the center of thinking and reasoning – are the seat of emotions and many innate behaviors as well as the control centers for muscles and automatic functions such as breathing.
Optical brain state switching
Dan, Weber and their colleagues chose a powerful technique called optogenetics to study these REM-related GABAergic neurons in the medulla. The technique involves inserting a light-sensitive ion channel into specific types of neurons by means of a virus. To target the virus to GABAergic neurons, the researchers used a genetically engineered mouse line that expresses a marker protein in these specific neurons only. Once present, the ion channel can turn on the activity of neurons when stimulated by laser light through an optical fiber inserted in the brain. Alternatively, inserting an inhibitory ion pump into the GABAergic neurons allowed the researchers to turn off the activity of these neurons through laser stimulation.
Using this genetically engineered strain of mice, the researchers mapped the activity of these neurons in the medulla and then recorded how activating or inactivating the neurons for brief periods affected sleep and waking behavior.
They also used a drug to inactivate the same set of neurons and found a reduction of REM sleep, though not as immediate and lasting for a longer period of time, since the drug required about half an hour to take effect and wore off slowly.
They also inserted the light-sensitive ion channels into a different set of neurons in the medulla: glutamatergic neurons, which release the neurotransmitter glutamate. Activating these neurons immediately awakened the animals, the opposite effect of activating the GABAergic neurons.
Dan is continuing her studies of the neurons that affect not only REM sleep, but also non-REM sleep.
Funding: The work was supported by Weber’s postdoctoral fellowships from the European Molecular Biology Organization and the Human Frontier Science Program. Other authors were Shinjae Chung and Min Xu of UC Berkeley and Kevin Beier and Liqun Luo of Stanford University.
Source: Robert Sanders – UC Berkeley
Image Credit: The image is credited to Franz Weber/UC Berkeley
Original Research: Abstract for “Control of REM sleep by ventral medulla GABAergic neurons” by Franz Weber, Shinjae Chung, Kevin T. Beier, Min Xu, Liqun Luo and Yang Dan in Nature. Published online October 7 2015 doi:10.1038/nature14979
Abstract
Control of REM sleep by ventral medulla GABAergic neurons
Rapid eye movement (REM) sleep is a distinct brain state characterized by activated electroencephalogram and complete skeletal muscle paralysis, and is associated with vivid dreams1, 2, 3. Transection studies by Jouvet first demonstrated that the brainstem is both necessary and sufficient for REM sleep generation2, and the neural circuits in the pons have since been studied extensively4, 5, 6, 7, 8. The medulla also contains neurons that are active during REM sleep9, 10, 11, 12, 13, but whether they play a causal role in REM sleep generation remains unclear. Here we show that a GABAergic (γ-aminobutyric-acid-releasing) pathway originating from the ventral medulla powerfully promotes REM sleep in mice. Optogenetic activation of ventral medulla GABAergic neurons rapidly and reliably initiated REM sleep episodes and prolonged their durations, whereas inactivating these neurons had the opposite effects. Optrode recordings from channelrhodopsin-2-tagged ventral medulla GABAergic neurons showed that they were most active during REM sleep (REMmax), and during wakefulness they were preferentially active during eating and grooming. Furthermore, dual retrograde tracing showed that the rostral projections to the pons and midbrain and caudal projections to the spinal cord originate from separate ventral medulla neuron populations. Activating the rostral GABAergic projections was sufficient for both the induction and maintenance of REM sleep, which are probably mediated in part by inhibition of REM-suppressing GABAergic neurons in the ventrolateral periaqueductal grey. These results identify a key component of the pontomedullary network controlling REM sleep. The capability to induce REM sleep on command may offer a powerful tool for investigating its functions.
“Control of REM sleep by ventral medulla GABAergic neurons” by Franz Weber, Shinjae Chung, Kevin T. Beier, Min Xu, Liqun Luo and Yang Dan in Nature. Published online October 7 2015 doi:10.1038/nature14979