Summary: Researchers report that, following a stroke, mapping the brain’s white matter connections in addition to imaging tissue damage could help to predict which patients will have language deficits and how severe they may be.
Source: Medical University of South Carolina.
Mapping damage to brain’s white matter connections after stroke can predict long-term language deficits, improve the understanding of how language is processed in the brain, and potentially inform the selection of rehabilitative therapy.
Loss or impairment of the ability to speak is one of the most feared complications of stroke–one faced by about 20% of stroke patients. Language, as one of the most complex functions of the brain, is not seated in a single brain region but involves connections between many regions.
In an article published in the June 22, 2016 issue of the Journal of Neuroscience, investigators at the Medical University of South Carolina (MUSC) and the University of South Carolina (USC) report that mapping all of the brain’s white matter connections after stroke, in addition to imaging the areas of cortical tissue damage, could better predict which patients will have language deficits and how severe those deficits will be. The totality of the brain’s connections is referred to as the connectome.
“Imaging the connectome of patients after stroke enables the identification of individual signatures of brain organization that can be used to predict the nature and severity of language deficits and one day could be used to guide therapy,” said MUSC Health neurologist Leonardo Bonilha M.D., Ph.D., senior author on the Journal of Neuroscience article, whose laboratory focuses on connectome imaging, particularly as it relates to language loss after stroke. Grigori Yourganov, Ph.D., is the first author on the article. Julius Fridriksson, Ph.D., Chris Rorden, Ph.D., and Ezequiel Gleichgerrcht, Ph.D, aphasia researchers at USC who recently received NIH funding to establish a Center for the Study of Aphasia Recovery and who are long-time collaborators of the Bonilha laboratory, are also authors on the article.
This study is the one of the first to use whole-brain connectome imaging to examine how disruptions to white matter connectivity after stroke affect language abilities. White matter fiber tracts are the insulated wires that connect one area of the brain to others. White matter is named for the myelin sheaths (insulation) that cover the many axons (wires) that make up the fiber tracts.
“If you have two brain areas and both of them have to work together in order to carry out a function and the stroke lesion takes out axons that connect those brain areas–the two areas are intact but the communication between them is disrupted and so there is dysfunction,” said Yourganov.
Currently, structural magnetic resonance imaging (MRI) is used after stroke to assess lesions in the cortical tissue–the brain’s grey matter. However, the extent of cortical damage often does not correlate with the severity of language deficits.
“Stroke patients sometimes have significant impairments beyond the amount of cortical damage,” said Bonilha. “It is also hard to predict how well a patient will recover based on the cortical lesion alone.”
Could connectome-based imaging be a useful complement for assessing damage to the brain’s connections after stroke and for guiding rehabilitative therapy?
The study led by Bonilha took an important first step toward answering these questions. The study, which enrolled 90 patients at MUSC and USC with aphasia due to a single stroke occurring no less than six months prior, assessed four areas related to speech/language using the Western Aphasia Battery–speech fluency, auditory comprehension, speech repetition, and oral naming–as well as a summary score of overall aphasia. Within two days of behavior assessment, each of the patients underwent imaging studies–both T1- and T2- weighted MRI, typically used after stroke to map cortical damage, and diffusion imaging, used for connectome mapping.
The team then used a type of machine learning algorithm–support vector regression (SVR)–to analyze the imaging results and make predictions about each patient’s language deficits. In essence, an algorithm was created that could derive the WAB score from either a feature relevant to imaging of the grey matter damage by structural MRI or a feature relevant to connectome imaging of the brain’s white matter fiber tracts. The team used 89 of the 90 patients as training sets for SVR and then used the algorithm to predict language defect/preservation in the 90th patient. This was done for each of the 90 patients and, in each patient, for both features identified via structural MRI and connectome imaging.
The accuracy of the algorithm’s prediction of WAB score for each patient was then assessed by comparing it to the WAB score determined via behavioral testing. Connectome-based analysis was as accurate as cortical lesion mapping for predicting WAB scores. In fact, it was better at predicting auditory comprehension scores than was lesion-based imaging using structural MRI and only slightly less accurate at predicting speech fluency, speech repetition, and naming scores.
The study demonstrates that damage to the white matter fiber tracts that connect the brain’s regions plays a role beyond cortical damage in language impairment after stroke. Furthermore, this study also discloses that connections in the brain’s parietal region are particularly important for language function, especially fluency. This region is less likely to sustain damage after stroke, even in patients who experience aphasia, suggesting that damage or preservation of the brain’s connections in this region could play a key role in determining who will experience aphasia and who will have the best chances for recovery. The integrity of these connections could not be mapped with conventional structural MRI but can now be assessed through connectome-based analysis.
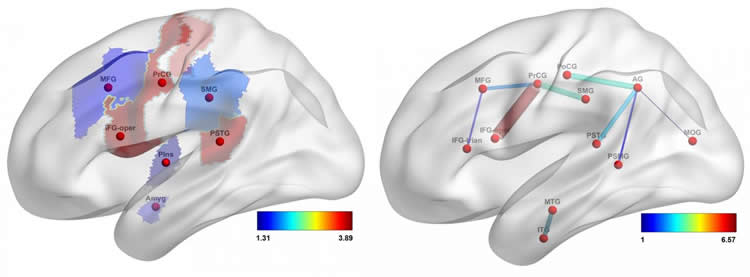
The study findings also suggest that connectome-based analysis could be used to inform a more individualized approach to stroke care. Because the algorithms developed using these study patients as the training set are generalizable to a broader stroke population, connectome-based analysis could one day be used to identify the distinctive features of each patient’s stroke–which connections have been lost and which preserved–and then the algorithm could be used to predict the type and severity of language impairment and the potential for recovery. This information could then be used to direct rehabilitative therapy to improve outcomes.
“By mapping much more accurately the individual pattern of brain structural connectivity in a stroke survivor, we can determine the integrity of neuronal networks and better understand what was lesioned and how that relates to language abilities that are lost,” said Bonilha. “This is, broadly stated, a measure of post-stroke brain health. It is the individual signature pattern that could also be used to inform about the personalized potential for recovery with therapy and guide treatments to focus on the deficient components of the network.”
Co-authors are Rice alumnus Benjamin Kirby and research scientist Christopher Pennington. Martí is an associate professor of chemistry, of bioengineering and of materials science and nanoengineering.
Funding: The NIH/National Institute on Deafness and Other Communication Disorders, and National Institutes of Health supported the research.
Source: Heather Woolwine – Medical University of South Carolina
Image Source: This NeuroscienceNews.com image is credited to Dr. Leonardo Bonilha and Dr. Grigori Yourganov of the Medical University of South Carolina.
Original Research:Abstract for “Multivariate Connectome-Based Symptom Mapping in Post-Stroke Patients: Networks Supporting Language and Speech” by Grigori Yourganov, Julius Fridriksson, Chris Rorden, Ezequiel Gleichgerrcht, and Leonardo Bonilha in Journal of Neuroscience. Published online June 22 2016 doi:10.1523/JNEUROSCI.4396-15.2016
[cbtabs][cbtab title=”MLA”]Medical University of South Carolina. “New Clue to How Lithium Works in the Brain.” NeuroscienceNews. NeuroscienceNews, 11 July 2016.
<https://neurosciencenews.com/language-stroke-connectome-4654/>.[/cbtab][cbtab title=”APA”]Medical University of South Carolina. (2016, July 11). New Clue to How Lithium Works in the Brain. NeuroscienceNews. Retrieved July 11, 2016 from https://neurosciencenews.com/language-stroke-connectome-4654/[/cbtab][cbtab title=”Chicago”]Medical University of South Carolina. “New Clue to How Lithium Works in the Brain.” https://neurosciencenews.com/language-stroke-connectome-4654/ (accessed July 11, 2016).[/cbtab][/cbtabs]
Abstract
Multivariate Connectome-Based Symptom Mapping in Post-Stroke Patients: Networks Supporting Language and Speech
Language processing relies on a widespread network of brain regions. Univariate post-stroke lesion-behavior mapping is a particularly potent method to study brain–language relationships. However, it is a concern that this method may overlook structural disconnections to seemingly spared regions and may fail to adjudicate between regions that subserve different processes but share the same vascular perfusion bed. For these reasons, more refined structural brain mapping techniques may improve the accuracy of detecting brain networks supporting language. In this study, we applied a predictive multivariate framework to investigate the relationship between language deficits in human participants with chronic aphasia and the topological distribution of structural brain damage, defined as post-stroke necrosis or cortical disconnection. We analyzed lesion maps as well as structural connectome measures of whole-brain neural network integrity to predict clinically applicable language scores from the Western Aphasia Battery (WAB). Out-of-sample prediction accuracy was comparable for both types of analyses, which revealed spatially distinct, albeit overlapping, networks of cortical regions implicated in specific aspects of speech functioning. Importantly, all WAB scores could be predicted at better-than-chance level from the connections between gray-matter regions spared by the lesion. Connectome-based analysis highlighted the role of connectivity of the temporoparietal junction as a multimodal area crucial for language tasks. Our results support that connectome-based approaches are an important complement to necrotic lesion-based approaches and should be used in combination with lesion mapping to fully elucidate whether structurally damaged or structurally disconnected regions relate to aphasic impairment and its recovery.
SIGNIFICANCE STATEMENT We present a novel multivariate approach of predicting post-stroke impairment of speech and language from the integrity of the connectome. We compare it with multivariate prediction of speech and language scores from lesion maps, using cross-validation framework and a large (n = 90) database of behavioral and neuroimaging data from individuals with post-stroke aphasia. Connectome-based analysis was similar to lesion-based analysis in terms of predictive accuracy and provided additional details about the importance of specific connections (in particular, between parietal and posterior temporal areas) for preserving speech functions. Our results suggest that multivariate predictive analysis of the connectome is a useful complement to multivariate lesion analysis, being less dependent on the spatial constraints imposed by underlying vasculature.
“Multivariate Connectome-Based Symptom Mapping in Post-Stroke Patients: Networks Supporting Language and Speech” by Grigori Yourganov, Julius Fridriksson, Chris Rorden, Ezequiel Gleichgerrcht, and Leonardo Bonilha in Journal of Neuroscience. Published online June 22 2016 doi:10.1523/JNEUROSCI.4396-15.2016







