Summary: Light sensitive cells in the fetal retina communicate as part of an interconnected network, giving the retina more light sensitivity during development that previously believed.
Source: UC Berkeley
By the second trimester, long before a baby’s eyes can see images, they can detect light.
But the light-sensitive cells in the developing retina — the thin sheet of brain-like tissue at the back of the eye — were thought to be simple on-off switches, presumably there to set up the 24-hour, day-night rhythms parents hope their baby will follow.
University of California, Berkeley, scientists have now found evidence that these simple cells actually talk to one another as part of an interconnected network that gives the retina more light sensitivity than once thought, and that may enhance the influence of light on behavior and brain development in unsuspected ways.
In the developing eye, perhaps 3% of ganglion cells — the cells in the retina that send messages through the optic nerve into the brain — are sensitive to light and, to date, researchers have found about six different subtypes that communicate with various places in the brain. Some talk to the suprachiasmatic nucleus to tune our internal clock to the day-night cycle. Others send signals to the area that makes our pupils constrict in bright light.
But others connect to surprising areas: the perihabenula, which regulates mood, and the amygdala, which deals with emotions.
In mice and monkeys, recent evidence suggests that these ganglion cells also talk with one another through electrical connections called gap junctions, implying much more complexity in immature rodent and primate eyes than imagined.
“Given the variety of these ganglion cells and that they project to many different parts of the brain, it makes me wonder whether they play a role in how the retina connects up to the brain,” said Marla Feller, a UC Berkeley professor of molecular and cell biology and senior author of a paper that appeared this month in the journal Current Biology. “Maybe not for visual circuits, but for non-vision behaviors. Not only the pupillary light reflex and circadian rhythms, but possibly explaining problems like light-induced migraines, or why light therapy works for depression.”
Parallel systems in developing retina
The cells, called intrinsically photosensitive retinal ganglion cells (ipRGCs), were discovered only 10 years ago, surprising those like Feller who had been studying the developing retina for nearly 20 years. She played a major role, along with her mentor, Carla Shatz of Stanford University, in showing that spontaneous electrical activity in the eye during development — so-called retinal waves — is critical for setting up the correct brain networks to process images later on.
Hence her interest in the ipRGCs that seemed to function in parallel with spontaneous retinal waves in the developing retina.
“We thought they (mouse pups and the human fetus) were blind at this point in development,” said Feller, the Paul Licht Distinguished Professor in Biological Sciences and a member of UC Berkeley’s Helen Wills Neuroscience Institute. “We thought that the ganglion cells were there in the developing eye, that they are connected to the brain, but that they were not really connected to much of the rest of the retina, at that point. Now, it turns out they are connected to each other, which was a surprising thing.”
UC Berkeley graduate student Franklin Caval-Holme combined two-photon calcium imaging, whole-cell electrical recording, pharmacology and anatomical techniques to show that the six types of ipRGCs in the newborn mouse retina link up electrically, via gap junctions, to form a retinal network that the researchers found not only detects light, but responds to the intensity of the light, which can vary nearly a billionfold.
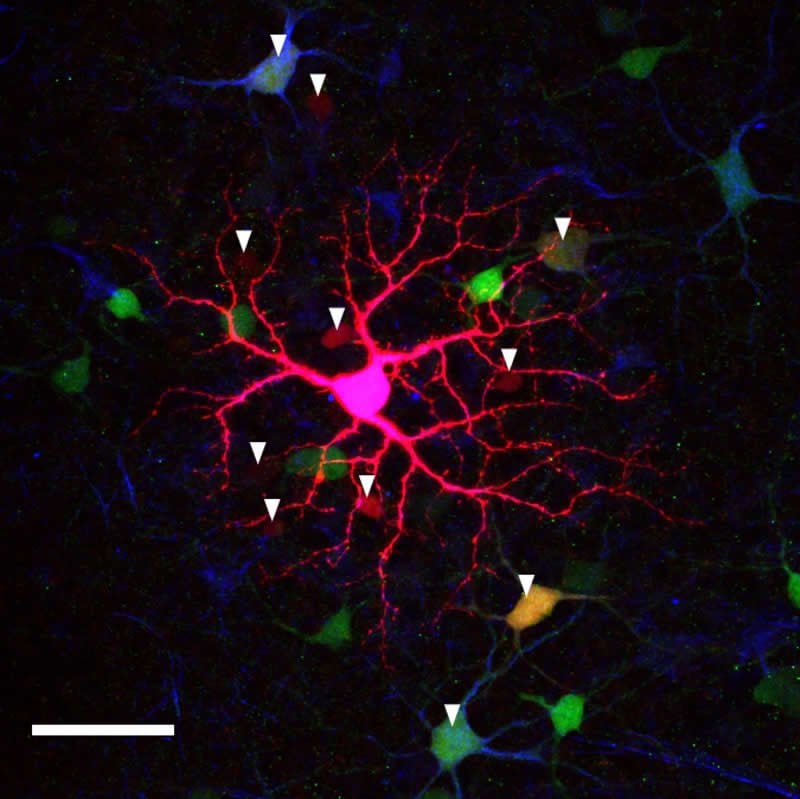
Gap junction circuits were critical for light sensitivity in some ipRGC subtypes, but not others, providing a potential avenue to determine which ipRGC subtypes provide the signal for specific non-visual behaviors that light evokes.
“Aversion to light, which pups develop very early, is intensity-dependent,” suggesting that these neural circuits could be involved in light-aversion behavior, Caval-Holme said. “We don’t know which of these ipRGC subtypes in the neonatal retina actually contributes to the behavior, so it will be very interesting to see what role all these different subtypes have.”
The researchers also found evidence that the circuit tunes itself in a way that could adapt to the intensity of light, which probably has an important role in development, Feller said.
“In the past, people demonstrated that these light-sensitive cells are important for things like the development of the blood vessels in the retina and light entrainment of circadian rhythms, but those were kind of a light on/light off response, where you need some light or no light,” she said. “This seems to argue that they are actually trying to code for many different intensities of light, encoding much more information than people had previously thought.”
Funding: The research was supported by the National Institutes of Health (NIH F31EY028022-03, RO1EY019498, RO1EY013528, P30EY003176).
Source:
UC Berkeley
Media Contacts:
Robert Sanders – UC Berkeley
Image Source:
The image is credited to Franklin Caval-Holme, UC Berkeley.
Original Research: Open access
“Gap Junction Coupling Shapes the Encoding of Light in the Developing Retina”. Franklin Caval-Holme, Marla B. Feller.
Current Biology doi:10.1016/j.cub.2019.10.025.
Abstract
Gap Junction Coupling Shapes the Encoding of Light in the Developing Retina
Highlights
• Six functional groups of light-responsive cells correspond to mixed ipRGC subtypes
• Gap junctions transmit slow photocurrents and spikelets
• Blocking gap junctions reduces light sensitivity in most functional groups
• Increased gap junction coupling enhances light sensitivity of all functional groups
Summary
Detection of ambient illumination in the developing retina prior to maturation of conventional photoreceptors is mediated by intrinsically photosensitive retinal ganglion cells (ipRGCs) and is critical for driving several physiological processes, including light aversion, pupillary light reflexes, and photoentrainment of circadian rhythms. The strategies by which ipRGCs encode variations in ambient light intensity at these early ages are not known. Using unsupervised clustering of two-photon calcium responses followed by inspection of anatomical features, we found that the population activity of the neonatal retina could be modeled as six functional groups that were composed of mixtures of ipRGC subtypes and non-ipRGC cell types. By combining imaging, whole-cell recording, pharmacology, and anatomical techniques, we found that functional mixing of cell types is mediated in part by gap junction coupling. Together, these data show that both cell-autonomous intrinsic light responses and gap junction coupling among ipRGCs contribute to the proper encoding of light intensity in the developing retina.






