Summary: Researchers report a promising treatment for reversing deafness, that converts inner ear stem cells to auditory neurons, may increase cancer risk as the process can make the cells divide too quickly.
Source: Rutgers University New Brunswick.
Want to restore hearing by injecting stem cells into the inner ear? Well, that can be a double-edged sword.
Inner ear stem cells can be converted to auditory neurons that could reverse deafness, but the process can also make those cells divide too quickly, posing a cancer risk, according to a study led by Rutgers University–New Brunswickscientists.
The encouraging news is that turning stem cells into auditory neurons can be controlled – at least in a Petri dish, said Kelvin Y. Kwan, senior author of the study and an assistant professor in the Department of Cell Biology and Neuroscience in the School of Arts and Sciences.
“It’s a cautionary tale,” Kwan said. “People say, ‘we’ll just put stem cells in and we’re going to replace lost neurons.’ We’re saying that ‘yes, we can make neurons,’ but you have other side effects that are unanticipated, such as increased proliferation of stem cells. So this will guide us toward a better strategy for cell replacement therapies.”
The Rutgers-led study was published in Stem Cell Reports and led by Zhichao Song, a doctoral student in Kwan’s lab. Co-author Azadeh Jadali is a post-doctoral associate in the lab.
So-called hair cells in the inner ear convert sounds into neural signals that are relayed to the brain by spiral ganglion neurons, the study notes. Hearing loss from overexposure to noise causes hair cell loss, severe damage to neuronal processes and slow degeneration of auditory neurons. The neurons do not regenerate once they are lost.
“Hearing loss impacts about 15 percent of the American population – probably more,” Kwan said. “Over the years, you don’t realize that you’re not hearing well until you get tested. We’re one of the few labs trying to figure out how to address the hearing loss issue.”
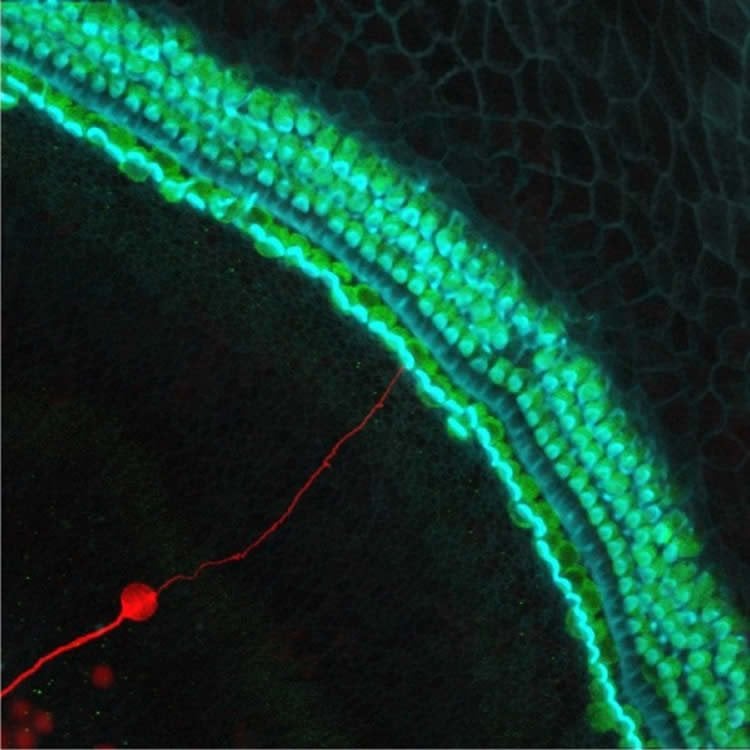
In their study, the scientists overexpressed a gene called NEUROG1 to turn inner ear stem cells into auditory neurons.
“But since that leads to increased cell division and NEUROG1 is used in other stem cells to make other types of neurons, scientists in other fields should be aware that when using this factor, they’ll probably also increase cell proliferation,” Kwan said.
The Rutgers scientists also discovered that chromatin – DNA studded with histone proteins – influences how NEUROG1 functions. Changes in chromatin may help reduce unwanted stem cell proliferation and can be achieved by adding drugs to experimental cultures in Petri dishes, Kwan said.
“Ideally, we would change the chromatin state before we start overexpressing NEUROG1 and prevent unwanted stem cell proliferation,” he said.
Source: Chris Melvin – Rutgers University New Brunswick
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Kelvin Y. Kwan/Rutgers University-New Brunswick
Original Research: Full open access research for “NEUROG1 Regulates CDK2 to Promote Proliferation in Otic Progenitors” by Zhichao Song, Azadeh Jadali, Bernd Fritzsch, and Kelvin Y. Kwan in Stem Cell Reports. Published online October 12 2017 doi:10.1016/j.stemcr.2017.09.011
[cbtabs][cbtab title=”MLA”]Rutgers University New Brunswick “Inner Ear Stem Cells May Someday Restore Hearing.” NeuroscienceNews. NeuroscienceNews, 7 November 2017.
<https://neurosciencenews.com/hearing-ear-stem-cells-7891/>.[/cbtab][cbtab title=”APA”]Rutgers University New Brunswick (2017, November 7). Inner Ear Stem Cells May Someday Restore Hearing. NeuroscienceNews. Retrieved November 7, 2017 from https://neurosciencenews.com/hearing-ear-stem-cells-7891/[/cbtab][cbtab title=”Chicago”]Rutgers University New Brunswick “Inner Ear Stem Cells May Someday Restore Hearing.” https://neurosciencenews.com/hearing-ear-stem-cells-7891/ (accessed November 7, 2017).[/cbtab][/cbtabs]
Abstract
NEUROG1 Regulates CDK2 to Promote Proliferation in Otic Progenitors
Highlights
•NEUROG1 is enriched at the Cdk2 promoter
•Chromatin at the Cdk2 promoter changes during proliferation and differentiation
•NEUROG1 promotes expression of CDK2 to drive otic progenitor proliferation
Summary
Loss of spiral ganglion neurons (SGNs) significantly contributes to hearing loss. Otic progenitor cell transplantation is a potential strategy to replace lost SGNs. Understanding how key transcription factors promote SGN differentiation in otic progenitors accelerates efforts for replacement therapies. A pro-neural transcription factor, Neurogenin1 (Neurog1), is essential for SGN development. Using an immortalized multipotent otic progenitor (iMOP) cell line that can self-renew and differentiate into otic neurons, NEUROG1 was enriched at the promoter of cyclin-dependent kinase 2 (Cdk2) and neurogenic differentiation 1 (NeuroD1) genes. Changes in H3K9ac and H3K9me3 deposition at the Cdk2 and NeuroD1 promoters suggested epigenetic regulation during iMOP proliferation and differentiation. In self-renewing iMOP cells, overexpression of NEUROG1 increased CDK2 to drive proliferation, while knockdown of NEUROG1 decreased CDK2 and reduced proliferation. In iMOP-derived neurons, overexpression of NEUROG1 accelerated acquisition of neuronal morphology, while knockdown of NEUROG1 prevented differentiation. Our findings suggest that NEUROG1 can promote proliferation or neuronal differentiation.
“NEUROG1 Regulates CDK2 to Promote Proliferation in Otic Progenitors” by Zhichao Song, Azadeh Jadali, Bernd Fritzsch, and Kelvin Y. Kwan in Stem Cell Reports. Published online October 12 2017 doi:10.1016/j.stemcr.2017.09.011






