Summary: A new analysis method shows the brain regulates its signals using glutamate in more ways than previously thought.
Source: Chalmers University of Technology
Researchers at Chalmers University of Technology and Gothenburg University in Sweden have achieved something long thought almost impossible – counting the molecules of the neurotransmitter glutamate released when a signal is transferred between two brain cells. With a new analysis method, they showed that the brain regulates its signals using glutamate in more ways than previously realised.
The ability to measure the activity and quantity of glutamate in brain cells has been long sought-after among researchers. Glutamate is the major excitatory neurotransmitter in the brain. Despite its abundance, and its influence on many important functions, we know a lot less about it than other neurotransmitters such as serotonin and dopamine, because so far glutamate has been difficult to measure quickly enough.
The new findings around glutamate are therefore very significant and could help improve our understanding of the pathologies underlying neurological and psychiatric diseases and conditions. The relationship between glutamate and these disorders, as well as our memory, our appetite and more, are just some of the questions which the researchers’ newly discovered technology could help answer.
“When we started, everybody said ‘this will never work’. But we didn’t give in. Now we have a beautiful example of how multi-disciplinary basic science can yield major breakthroughs, and deliver real benefit,” says Ann-Sofie Cans, Associate Professor in Chemistry at Chalmers and leader of the research group.
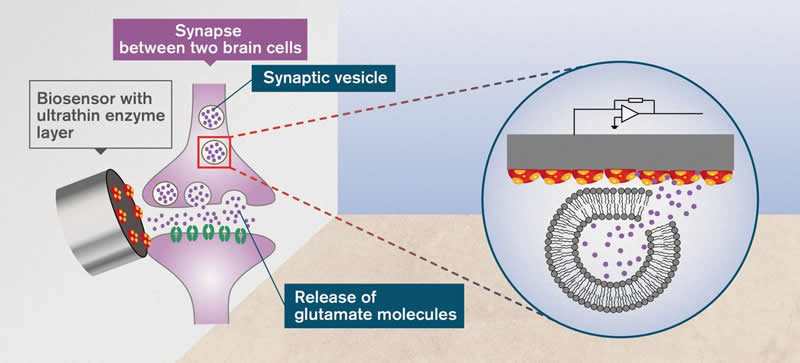
The key was to do the opposite of what had been previously attempted. Instead of using a biosensor made from thick layers, they used an ultrathin layer of the enzyme needed for biological identification. The researchers made it so that the enzyme, which was placed on a nano-structured sensor surface, was just a molecule thick. This made the sensor technology a thousand times faster than previous attempts.
The technique was therefore fast enough to measure the release of glutamate from a single synaptic vesicle – the small liquid vessel which releases neurotransmitters to the synapse between two nerve cells. This is a process that occurs in less than a thousandth of a second.
“When we saw the benefits of improving the sensor technology in terms of time, instead of concentration, then we got it to work” says Ann-Sofie Cans.
The research was carried out in two steps. In the first, the breakthrough was being able to measure glutamate. That study was published early in Spring 2019 in the scientific journal ASC Chemical Neuroscience. In the second part, which the current publication addresses, Ann-Sofie Cans and her research group made further important adjustments and ground-breaking discoveries.
“Once we had built the sensor, we could then refine it further. Now, with the help of this technology we have also developed a new method to quantify these small amounts of glutamate,” she explains.
Along the way the group had many interesting surprises. For example, the quantity of glutamate in a synaptic vesicle has been revealed to be much greater than previously believed. It is comparable in quantity to serotonin and dopamine, a finding which came as an exciting surprise.
“Our study changes the current understanding of glutamate. For example, it seems that transport and storage of glutamate in synaptic vesicles is not as different as we thought, when compared with other neurotransmitters like serotonin and dopamine”, says Ann-Sofie Cans.
The researchers also showed that nerve cells control the strength of their chemical signals by regulating the quantity of glutamate released from single synaptic vesicles.
The fact that researchers can now measure and quantify this neurotransmitter can yield new tools for pharmacological studies in many vital areas in neuroscience.
“The level of measurement offered by this ultra-fast glutamate sensor opens up countless possibilities to truly understand the function of glutamate in health and disease. Our knowledge of the brain function, and dysfunction, is limited by the experimental tools we have, and this new ultra-fast tool will allow us to examine neuronal communication at a level we did not have access to before”, says Karolina Patrycja Skibicka, Associate Professor in Neuroscience and Physiology at Gothenburg University.
“The new finding, that glutamate-based communication is regulated by the quantity of glutamate released from synaptic vesicles, begs the question of what happens to this regulation in brain diseases thought to be linked to glutamate, for example epilepsy.”
More information on glutamate and glutamic acid:
Glutamate, or glutamic acid, is found in proteins in food. It occurs naturally in meat, in almost all vegetables, and in wheat and soy. It is also used as a food additive to enhance flavours, for example in the form of MSG, or monosodium glutamate.
Glutamate is an amino acid, and an important part of our body. It is also a neurotransmitter which nerve cells use to communicate, and forms the basis for some of the brain’s basic functions such as cognition, memory and learning. It is also important for the immune system, the function of the gastrointestinal tract, and to prevent microorganisms from entering the body.
Source: Swedish Food Agency and Chalmers University of Technology
Source:
Chalmers University of Technology
Media Contacts:
Johanna Wilde – Chalmers University of Technology
Image Source:
The image is credited to Chalmers University of Technology.
Original Research: Closed access
“Counting the Number of Glutamate Molecules in Single Synaptic Vesicles”. Yuanmo Wang, Hoda Fathali, Devesh Mishra, Thomas Olsson, Jacqueline D. Keighron, Karolina P. Skibicka, Ann-Sofie Cans.
Journal of the American Chemical Society doi:10.1021/jacs.9b09414.
Abstract
Counting the Number of Glutamate Molecules in Single Synaptic Vesicles
Analytical tools for quantitative measurements of glutamate, the principal excitatory neurotransmitter in the brain, are lacking. Here, we introduce a new enzyme-based amperometric sensor technique for the counting of glutamate molecules stored inside single synaptic vesicles. In this method, an ultra-fast enzyme-based glutamate sensor is placed into a solution of isolated synaptic vesicles, which stochastically rupture at the sensor surface in a potential-dependent manner at a constant negative potential. The continuous amperometric signals are sampled at high speed (10 kHz) to record sub-millisecond spikes, which represent glutamate release from single vesicles that burst open. Glutamate quantification is achieved by a calibration curve that is based on measurements of glutamate release from vesicles pre-filled with various glutamate concentrations. Our measurements show that an isolated single synaptic vesicle encapsulates about 8000 glutamate molecules and is comparable to the measured exocytotic quantal glutamate release in amperometric glutamate sensing in the nucleus accumbens of mouse brain tissue. Hence, this new methodology introduces the means to quantify ultra-small amounts of glutamate and to study synaptic vesicle physiology, pathogenesis, and drug treatments for neuronal disorders where glutamate is involved.






