Last summer, a 74-year-old resident of San Francisco’s Chinatown was admitted to San Francisco General Hospital and Trauma Center with fever and disorientation, which rapidly degenerated into a coma.
Her symptoms screamed that she was suffering from encephalitis, an infection of the brain, but a series of tests and drugs – for tuberculosis, bacterial and fungal infections, even Toxoplasmosis – could neither identify the cause of infection nor do anything to resuscitate her.
She died 16 days later, her illness still a mystery to her clinicians.
Sadly, this outcome is all too common in cases of brain infection, which go unsolved six times out of 10 in California, according to a 2006 UC San Francisco study.
Biochemist Joe DeRisi, PhD and neurologist Michael Wilson, MD, of UCSF want to change that statistic, and they think they have the tools to do it.
When the researchers learned of last summer’s case of mystery encephalitis, they turned to cutting-edge genomic tools to track down the cause of the woman’s disease.
They used rapid next-generation genomic sequencing to search the patient’s spinal fluid for non-human DNA, then compared what they found against databases of genes of every known invasive microorganism or virus. This “metagenomic” approach rapidly found a match: a common soil-dwelling amoeba called Balamuthia mandrillaris, as the researchers reported last month in the journal Annals of Neurology.
The researchers and their colleagues have recently set up a new Center for Next-Gen Precision Medicine Diagnostics to help hospitals make genome-based screens for the root causes of encephalitis part of their standard of care.
The technology could save money as well as lives. Over her two-week hospitalization, care for last summer’s Balamuthia victim cost more than $100,000. The metagenomic screen, by comparison, costs a mere $2,000 – and typically takes two days.
One of the center’s first priorities will be a study to determine when in the course of an encephalitis patient’s care the technique would have the greatest clinical impact. Researchers will comb through databases of encephalitis cases to identify when cheap, existing treatments like a $100 blood draw are effective at diagnosing a patient’s disease and when it’s time for clinicians to turn to the center’s big genomic guns.
“We all agree these metagenomic screens are happening too late,” said UC Berkeley health economist Brent Fulton, PhD, MBA, part of the center’s team who is leading the study. “When UCSF gets the call, doctors have already tried everything under the sun and it’s a bad situation.”
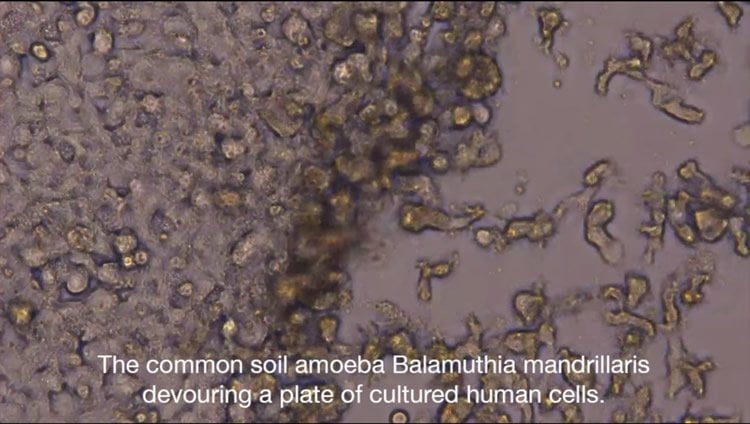
Balamuthia mandrillaris chows down on a plate of cultured human cells.
But how early should the test be done? It’s still an open question, according to Fulton. “A $2,000 test is nothing to sneeze at if it turns out to not change the usual course of care,” he explained. “But balanced against a multi-week, half-million dollar hospitalization, it’s nothing.”
Through the new center, DeRisi hopes to make such screening a hospital’s go-to test for mystery brain infections rather than a last resort. Currently, doctors need to track down DeRisi and his peers directly to request help, he said. “We need to make this into an actual clinical diagnostic so a doctor who doesn’t know my phone number can just order up this test.”
Source: Nicholas Weiler – UCSF
Image Source: The image is adapted from the UCSF video and is credited to the researchers
Video Source: The video is available at the UCSF YouTube page
Original Research: Full open access research for “Diagnosing Balamuthia mandrillaris Encephalitis With Metagenomic Deep Sequencing” by Michael R. Wilson MD, Niraj M. Shanbhag MD, PhD, Michael J. Reid MD, Neel S. Singhal MD, PhD, Jeffrey M. Gelfand MD, MAS, Hannah A. Sample BS, Barlas Benkli BA, Brian D. O’Donovan MS, Ibne K.M. Ali PhD, M. Kelly Keating DVM, Thelma H. Dunnebacke PhD, Matthew D. Wood MD, PhD, Andrew Bollen MD and Joseph L. DeRisi PhD in Annals of Neurology. Published online August 24 2015 doi:10.1002/ana.24499
Abstract
Diagnosing Balamuthia mandrillaris Encephalitis With Metagenomic Deep Sequencing
Meningoencephalitis presents significant diagnostic challenges owing, in part, to the large number of possible underlying causes. These include infectious, inflammatory, paraneoplastic, and toxic-metabolic etiologies, many of which can present with similar clinical phenotypes. Furthermore, confirming an infectious etiology can be challenging, given that many pathogen-specific serological and polymerase chain reaction (PCR) assays are expensive, time-consuming, and have poor operating characteristics. Culture-based approaches, though they can often detect multiple species, are also limited, given that many infectious agents require special culture conditions and prolonged incubation times. Standard culture protocols are also not commercially available for many organisms.
“Diagnosing Balamuthia mandrillaris Encephalitis With Metagenomic Deep Sequencing” by Michael R. Wilson MD, Niraj M. Shanbhag MD, PhD, Michael J. Reid MD, Neel S. Singhal MD, PhD, Jeffrey M. Gelfand MD, MAS, Hannah A. Sample BS, Barlas Benkli BA, Brian D. O’Donovan MS, Ibne K.M. Ali PhD, M. Kelly Keating DVM, Thelma H. Dunnebacke PhD, Matthew D. Wood MD, PhD, Andrew Bollen MD and Joseph L. DeRisi PhD in Annals of Neurology. Published online August 24 2015 doi:10.1002/ana.24499







