Scientists at the University of Copenhagen have captured thousands of progenitor cells of the pancreas on video. They have filmed the cells making decisions to either divide and expand the organ or to specialize into the endocrine cells that regulate our blood sugar levels.
The new study reveals that stem cells behave as people in a society, making individual choices but with enough interactions to bring them to their end-goal. The results could eventually lead to a better control over the production of insulin-producing endocrine cells for diabetes therapy.
The research is published in the scientific journal PLOS Biology.
Why one cell matters
In a joint collaboration between the University of Copenhagen and University of Cambridge, Professor Anne Grapin- Botton and a team of researchers including Assistant Professor Yung Hae Kim from danstem.ku.dk focused on marking the progenitor of the embryonic pancreas, commonly referred to as ‘mothers’, and their ‘daughters’ in different fluorescent colors and then captured them on video to analyze how they make decisions.
Prior to this work, there were methods to predict how specific types of pancreas cells would evolve as the embryo develops. However, by looking at individual cells, the scientists found that even within one group of cells presumed to be of the same type, some will divide many times to make the organ bigger while others will become specialized and will stop dividing.
The scientists witnessed interesting occurrences where the ‘mother’ of two ‘daughters’ made a decision and passed it on to the two ‘daughters’ who then acquired their specialization in synchrony. By observing enough cells, they were able to extract logic rules of decision-making, and with the help of Dr Pau Rué, a mathematician from the University of Cambridge, they developed a mathematical model to make long-term predictions over multiple generations of cells.
Stem cell movies
“It is the first time we have made movies of a quality that is high enough to follow thousands of individual cells in this organ, for periods of time that are long enough for us to follow the slow decision process. The task seemed daunting and technically challenging, but fascinating,” says Professor Grapin-Botton. “With these movies we can see and quantify the dynamics of decisions in each cell in the context of the organ, in a way that will inspire the study of many other organs,” says Assistant Professor Yung Hae Kim.
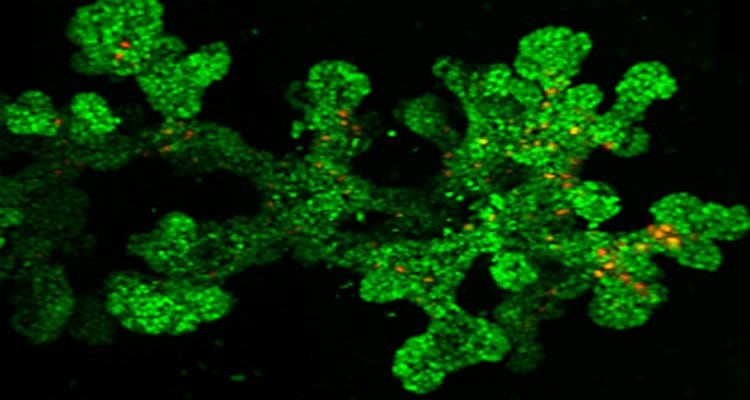
“To complement the movies, which are done on isolated pancreas, we developed a method to visualize the family tree of cells in the untouched organ. We initially focused on one generation but now we are aslo observing their descendants over multiple generations,” Kim elaborates.
Next steps in diabetes therapy
The project has been focused on basic research and is highly theoretical, but it now provides tools to control whether a cell should expand or specialize into an endocrine cell on its way to producing insulin. ‘It is a worldwide quest to produce such insulin-producing cells from stem cells, for their transplantation in diabetic patients. In the future, this could be done by increasing the probability of specialization or by pushing ‘mother’ cells to pass on the decision to specialize to their two daughters’, Grapin-Botton concludes.
Contact: Yung Hae Kim – University of Copenhagen
Source: University of Copenhagen press release
Image Source: The image is credited to the researchers and is adapted from the University of Copenhagen press release
Original Research: Full open access research for “Cell Cycle–Dependent Differentiation Dynamics Balances Growth and Endocrine Differentiation in the Pancreas” by Yung Hae Kim, and Hjalte List Larsen, Pau Rué, Laurence A. Lemaire, Jorge Ferrer, Anne Grapin-Botton in PLOS Biology. Published online March 18 2015 doi:10.1371/journal.pbio.1002111
Cell Cycle–Dependent Differentiation Dynamics Balances Growth and Endocrine Differentiation in the Pancreas
Organogenesis relies on the spatiotemporal balancing of differentiation and proliferation driven by an expanding pool of progenitor cells. In the mouse pancreas, lineage tracing at the population level has shown that the expanding pancreas progenitors can initially give rise to all endocrine, ductal, and acinar cells but become bipotent by embryonic day 13.5, giving rise to endocrine cells and ductal cells. However, the dynamics of individual progenitors balancing self-renewal and lineage-specific differentiation has never been described. Using three-dimensional live imaging and in vivo clonal analysis, we reveal the contribution of individual cells to the global behaviour and demonstrate three modes of progenitor divisions: symmetric renewing, symmetric endocrinogenic, and asymmetric generating a progenitor and an endocrine progenitor. Quantitative analysis shows that the endocrine differentiation process is consistent with a simple model of cell cycle–dependent stochastic priming of progenitors to endocrine fate. The findings provide insights to define control parameters to optimize the generation of β-cells in vitro.
“Cell Cycle–Dependent Differentiation Dynamics Balances Growth and Endocrine Differentiation in the Pancreas” by Yung Hae Kim, and Hjalte List Larsen, Pau Rué, Laurence A. Lemaire, Jorge Ferrer, Anne Grapin-Botton in PLOS Biology doi:10.1371/journal.pbio.1002111.






