Summary: A new study reveals, in rodents, some spinal pain begins in the frontal cortex. Researchers say the findings could help in the development of new treatments for chronic pain.
Source: University of Toronto.
A University of Toronto scientist has discovered the brain’s frontal lobe is involved in pain transmission to the spine. If his findings in animals bear out in people, the discovery could lead to a new class of non-addictive painkillers.
For 20 years, Min Zhuo, a professor of physiology in the Faculty of Medicine, has been intrigued by invisible pain, in particular chronic pain with no obvious cause. He has long suspected that the standard way of viewing spinal pain – it must come from injury or tissue inflammation – and the usual understanding of how to treat it – block it from entering the spinal cord – wasn’t telling the whole story.
Now, Zhuo has shown in mice and rats that some spinal pain actually begins in the brain’s frontal lobe, an area previously thought to be uninvolved. And he has shown how treating the pain in this area could be effective at preventing chronic pain. Zhuo published his results May 16 in the journal Nature Communication.
“When doctors can’t see anything wrong to cause chronic pain, often they think patients are making it up,” says Zhuo. “But pain that originates in the frontal lobe would be very different from pain that comes from a physical injury, like a herniated disc. There wouldn’t necessarily be any injury to see. That’s because our personality and emotions live in this region. If the frontal lobe can produce physical pain, that pain would be deeply tied to emotions like anxiety.”
Scientists already knew that the prefrontal cortex was in some way involved in pain because it would light up in scans of people in pain. But that activity was always thought to be symptom not cause, says Zhuo.
“When you have extreme anxiety, more neurotransmitters are released that end up causing pain in the spine,” he says. “Normal functions like walking shouldn’t be painful. But this flood of neurotransmitters sends the spine into hyper drive, and it starts treating ordinary sensations like pain. That could explain why anxiety can cause chest pain and make you think you’re having a heart attack. Or why some people experience pain when you touch them. I believe this helps to explain why emotional pain causes physical pain.”
The good news is that pain from the frontal lobe seems to be transmitted in a simple, direct way to the spine – making it relatively easy to shut down. Neurons in the frontal cortex send signals all the way down the spinal cord, says Zhuo, whereas pain signals from other areas of the brain are mediated by a complex network.
In animals, Zhuo found that pain was associated with increased neurotransmitters released from the frontal cortex. He was able to lessen pain by reducing the amount released. His next step is to test this process in people.
Those who suffer from anxiety along with neuropathic pain would likely benefit from a painkiller targeting the frontal lobe, says Zhuo.
Funding: The research was supported by the National Key Basic Science Research Project and the Natural Science Foundation of China.
Source: Heidi Singer – University of Toronto
Publisher: Organized by NeuroscienceNews.com.
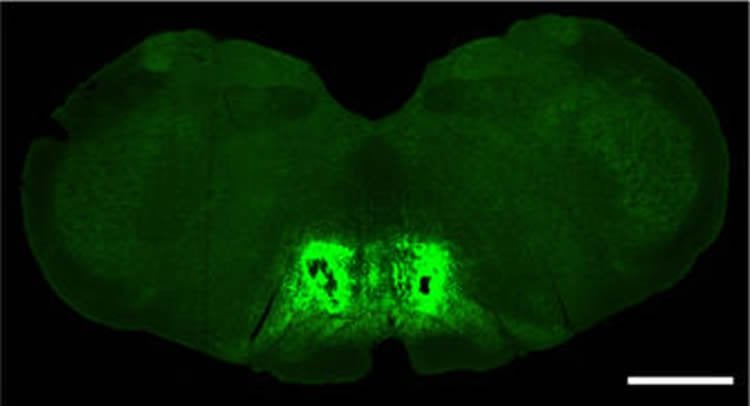
Image Source: NeuroscienceNews.com image is credited to Zhuo et al./Nature Communications.
Original Research: Open access research for “Top-down descending facilitation of spinal sensory excitatory transmission from the anterior cingulate cortex” by Tao Chen, Wataru Taniguchi, Qi-Yu Chen, Hidetoshi Tozaki-Saitoh, Qian Song, Ren-Hao Liu, Kohei Koga, Tsuyoshi Matsuda, Yae Kaito-Sugimura, Jian Wang, Zhi-Hua Li, Ya-Cheng Lu, Kazuhide Inoue, Makoto Tsuda, Yun-Qing Li, Terumasa Nakatsuka & Min Zhuo in Nature Communications. Published May 14 2018.
doi:10.1038/s41467-018-04309-2
[cbtabs][cbtab title=”MLA”]University of Toronto “Frontal Lobe Implicated in Chronic Pain.” NeuroscienceNews. NeuroscienceNews, 26 May 2018.
<https://neurosciencenews.com/frontal-lobe-chronic-pain-9148/>.[/cbtab][cbtab title=”APA”]University of Toronto (2018, May 26). Frontal Lobe Implicated in Chronic Pain. NeuroscienceNews. Retrieved May 26, 2018 from https://neurosciencenews.com/frontal-lobe-chronic-pain-9148/[/cbtab][cbtab title=”Chicago”]University of Toronto “Frontal Lobe Implicated in Chronic Pain.” https://neurosciencenews.com/frontal-lobe-chronic-pain-9148/ (accessed May 26, 2018).[/cbtab][/cbtabs]
Abstract
Top-down descending facilitation of spinal sensory excitatory transmission from the anterior cingulate cortex
Spinal sensory transmission is under descending biphasic modulation, and descending facilitation is believed to contribute to chronic pain. Descending modulation from the brainstem rostral ventromedial medulla (RVM) has been the most studied, whereas little is known about direct corticospinal modulation. Here, we found that stimulation in the anterior cingulate cortex (ACC) potentiated spinal excitatory synaptic transmission and this modulation is independent of the RVM. Peripheral nerve injury enhanced the spinal synaptic transmission and occluded the ACC-spinal cord facilitation. Inhibition of ACC reduced the enhanced spinal synaptic transmission caused by nerve injury. Finally, using optogenetics, we showed that selective activation of ACC-spinal cord projecting neurons caused behavioral pain sensitization, while inhibiting the projection induced analgesic effects. Our results provide strong evidence that ACC stimulation facilitates spinal sensory excitatory transmission by a RVM-independent manner, and that such top-down facilitation may contribute to the process of chronic neuropathic pain.