Summary: Researchers have revealed the 3d structure of the Flycatcher1 protein channel which enables the Venus flytrap to close in response to prey. The findings shed light on how other mechanosensitive ion channels may operate in other plants, bacteria, and the human body.
Source: Scripps Research Institute
Scientists at Scripps Research have revealed the three-dimensional structure of Flycatcher1, an aptly named protein channel that may enable Venus fly trap plants to snap shut in response to prey.
The structure of Flycatcher1, published February 14 in Nature Communications, helps shed light on longstanding questions about the remarkably sensitive touch response of Venus fly traps.
The structure also gives the researchers a better understanding of how similar proteins in organisms including plants and bacteria, as well as proteins in the human body with similar functions (called mechanosensitive ion channels), might operate.
“Despite how different Venus fly traps are from humans, studying the structure and function of these mechanosensitive channels gives us a broader framework for understanding the ways that cells and organisms respond to touch and pressure,” says co-senior author and Scripps Research professor Andrew Ward, PhD.
“Every new mechanosensitive channel that we study helps us make progress in understanding how these proteins can sense force and translate that to action and ultimately reveal more about human biology and health,” adds co-senior author Ardem Patapoutian, PhD, a Scripps Research professor who won the Nobel Prize in Physiology or Medicine for research on the mechanosensitive channels that allow the body to sense touch and temperature.
Mechanosensitive ion channels are like tunnels that span the membranes of cells. When jostled by movement, the channels open, letting charged molecules rush across. In response, cells then alter their behavior—a neuron might signal its neighbor, for instance.
The ability for cells to sense pressure and movement is important for people’s senses of touch and hearing, but also for many internal body processes—from the ability of the bladder to sense that it’s full to the ability of lungs to sense how much air is being breathed.
Previously, scientists had homed in on three ion channels in Venus fly traps thought to be related to the ability of the carnivorous plant to snap its leaves shut when its sensitive trigger hairs get touched. One, Flycatcher1, caught researchers’ attention because its genetic sequence looked similar to a family of mechanosensitive channels, MscS, found in bacteria.
“The fact that variants of this channel are found throughout evolution tells us that it must have some fundamental, important functions that have been maintained in different types of organisms,” says co-first author Sebastian Jojoa-Cruz, a graduate student at Scripps Research.
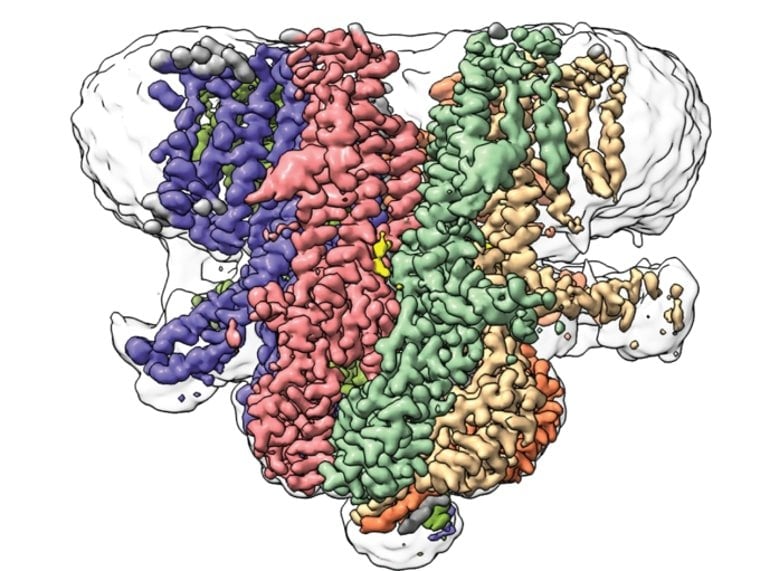
In the new study, the researchers used cryo-electron microscopy—a cutting-edge technique that reveals the locations of atoms within a frozen protein sample—to analyze the precise arrangement of molecules that form the Flycatcher1 protein channel in Venus fly trap plants. They found that Flycatcher1 is, in many ways, similar to bacterial MscS proteins—seven groups of identical helices surrounding a central channel.
But, unlike other MscS channels, Flycatcher1 has an unusual linker region extending outward from each group of helices. Like a switch, each linker can be flipped up or down. When the team determined the structure of Flycatcher1, they found six linkers in the down position, and just one flipped up.
“The architecture of Flycatcher1’s channel core was similar to other channels that have been studied for years, but these linker regions were surprising,” says Kei Saotome, PhD, a former postdoctoral research associate at Scripps Research and co-first author of the new paper.
To help elucidate the function of these switches, the researchers altered the linker to disrupt the up position. Flycatcher1, they found, no longer functioned as usual in response to pressure; the channel remained open for a longer duration when it would normally close upon removal of pressure.
“The profound effect of this mutation tells us that the conformations of these seven linkers is likely relevant for how the channel works,” says co-senior author Swetha Murthy, PhD, of Vollum Institute at Oregon Health and Science University, a former postdoctoral research associate at Scripps Research.
Now that they solved the molecular structure, the research team is planning future studies on the function of Flycatcher1 to understand how different conformations affect its function. More work is also needed to determine whether Flycatcher1 is solely responsible for the snapping shut of Venus fly trap leaves, or whether other suspected channels play complementary roles.
In addition to Jojoa-Cruz, Saotome, Murthy, Patapoutian and Ward, authors of the study, “Structural insights into the Venus flytrap mechanosensitive ion channel Flycatcher1,” are Che Chun Alex Tsui and Wen-Hsin Lee of Scripps Research, and Mark Sansom of University of Oxford.
Funding: This work and the researchers involved were supported by funding from the National Institutes of Health (R01 HL143297, R01 HL143297), a Ray Thomas Edwards Foundation grant, the Wellcome Trust (grant 208361/Z/17/Z), the Biotechnology and Biological Sciences Research Council (grants BB/N000145/1 and BB/R00126X/1), the Engineering and Physical Sciences Research Council (grant EP/R004722/1), a postdoctoral fellowship from the Jane Coffin Childs Memorial Fund for Medical Research, the Skaggs-Oxford Scholarship, the Croucher Foundation, and the Howard Hughes Medical Institute.
About this neuroscience research news
Author: Press Office
Source: Scripps Research Institute
Contact: Press Office – Scripps Research Institute
Image: The image is credited to Scripps Research Institute
Original Research: Open access.
“Structural insights into the Venus flytrap mechanosensitive ion channel Flycatcher1” by Ardem Patapoutian et al. Nature Communications
Abstract
Structural insights into the Venus flytrap mechanosensitive ion channel Flycatcher1
Flycatcher1 (FLYC1), a MscS homolog, has recently been identified as a candidate mechanosensitive (MS) ion channel involved in Venus flytrap prey recognition. FLYC1 is a larger protein and its sequence diverges from previously studied MscS homologs, suggesting it has unique structural features that contribute to its function.
Here, we characterize FLYC1 by cryo-electron microscopy, molecular dynamics simulations, and electrophysiology. Akin to bacterial MscS and plant MSL1 channels, we find that FLYC1 central core includes side portals in the cytoplasmic cage that regulate ion preference and conduction, by identifying critical residues that modulate channel conductance.
Topologically unique cytoplasmic flanking regions can adopt ‘up’ or ‘down’ conformations, making the channel asymmetric. Disruption of an up conformation-specific interaction severely delays channel deactivation by 40-fold likely due to stabilization of the channel open state.
Our results illustrate novel structural features and likely conformational transitions that regulate mechano-gating of FLYC1.






