Summary: Findings reveal a role the endocannabinoid system plays in a range of psychiatric disorders, including schizophrenia, bipolar disorder, and ASD.
Source: Northwestern University
Northwestern Medicine scientists discovered an unexpected connection between a synapse protein that has been implicated in neuropsychiatric disorders and the endocannabinoid pathway, according to a study published in Biological Psychiatry.
These findings suggest a role for the endocannabinoid system in conditions including bipolar disorder, according to Peter Penzes, PhD, the Ruth and Evelyn Dunbar Professor of Psychiatry and Behavioral Sciences, professor of Physiology and Pharmacology, and senior author of the study.
“The endocannabinoid system could be disrupted in patients with bipolar disease, or it could be the opposite: medical marijuana could have therapeutic potential for these patients,” said Penzes, who is also director of the Center for Autism and Neurodevelopment. “These are the questions that need to be answered.”
Cannabis has an effect on humans because it mimics endocannabinoids, chemicals occurring naturally in the brain. While the specific function of endocannabinoids is still not fully understood, the legalization of marijuana in many U.S. states has prompted more investigation into its biological pathways, Penzes said.
Endocannabinoids are produced by an enzyme called diacylglycerol lipase alpha (DAGLA), which is concentrated in synapses. Endocannabinoids dampen synaptic strength, one reason for the calming effects of marijuana.
Penzes and his collaborators have previously studied ankyrin-G, another synapse protein that regulated the speed of transmission across synapses. Aberrant expression of ankyrin-G — either too much or too little — has been associated with disorders such as bipolar disorder, schizophrenia and autism.
Studying mice genetically modified to lack ankyrin-G, they made a surprising discovery: Ankyrin-G appeared to stabilize DAGLA at synapses, making DAGLA more efficient.
“It’s a delicate mechanism that regulates dendritic spine morphology,” said Sehyoun Yoon, PhD, research assistant professor of Physiology and lead author of the study.
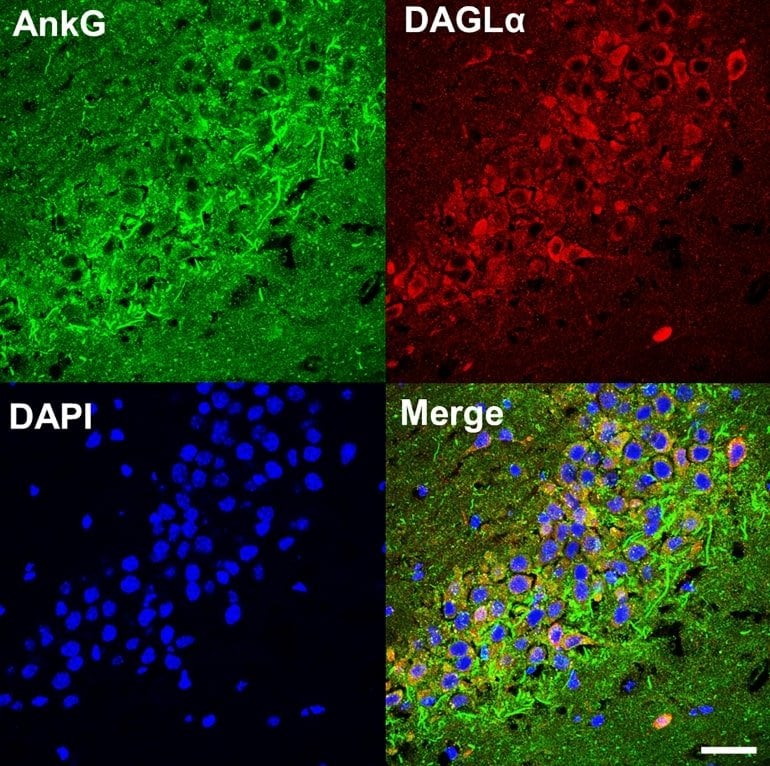
These findings comport with another recent study, led by investigators at Icahn School of Medicine at Mount Sinai and published in Nature Genetics. The study showed that both DAGLA and ankyrin-G (ANK3) are risk genes for bipolar disorder in a genome analysis of over 40,000 patients.
“It’s almost like somebody who is leading a double life, Dr. Jekyll and Mr. Hyde,” Penzes said. “Ankyrin-G has this entire separate function.”
The convergence of ankyrin-G with the endocannabinoid pathway opens up an entire new world of possibilities, both for investigating disease risk and possible therapies.
“Cannabis may contribute to increased risk for mental disorders, which has actually been shown in schizophrenia,” Penzes said. “Conversely, cannabis could be beneficial in some brain disorders, which prompted trials of medical marijuana in patients with autism.”
In the future, Penzes said he plans to examine the downstream effects of this biological pathway, both in normal subjects and in disease.
Funding: This work was supported by the National Institute of Mental Health grant R01MH107182.
About this mental health research news
Source: Northwestern University
Contact: Marla Paul – Northwestern University
Image: The image is credited to the researchers
Original Research: Closed access.
“cAMP Signaling–Mediated Phosphorylation of Diacylglycerol Lipase α Regulates Interaction With Ankyrin-G and Dendritic Spine Morphology” by Peter Penzes et al. Biological Psychiatry
Abstract
cAMP Signaling–Mediated Phosphorylation of Diacylglycerol Lipase α Regulates Interaction With Ankyrin-G and Dendritic Spine Morphology
Background
Diacylglycerol lipase α (DAGLα), a major biosynthetic enzyme for endogenous cannabinoid signaling, has emerged as a risk gene in multiple psychiatric disorders. However, its role in the regulation of dendritic spine plasticity is unclear.
Methods
DAGLα wild-type or point mutants were overexpressed in primary cortical neurons or human embryonic kidney 293T cells. The effects of mutated variants on interaction, dendritic spine morphology, and dynamics were examined by proximity ligation assay or fluorescence recovery after photobleaching. Behavioral tests and immunohistochemistry were performed with ankyrin-G conditional knockout and wild-type male mice.
Results
DAGLα modulated dendritic spine size and density, but the effects of changes in its protein level versus enzymatic activity were different, implicating either a 2-arachidonoylglycerol (2-AG)–dependent or –independent mechanism. The 2-AG–independent effects were mediated by the interaction of DAGLα with ankyrin-G, a multifunctional scaffold protein implicated in psychiatric disorders. Using superresolution microscopy, we observed that they colocalized in distinct nanodomains, which correlated with spine size. In situ proximity ligation assay combined with structured illumination microscopy revealed that DAGLα phosphorylation upon forskolin treatment enhanced the interaction with ankyrin-G in spines, leading to increased spine size and decreased DAGLα surface diffusion. Ankyrin-G conditional knockout mice showed significantly decreased DAGLα-positive neurons in the forebrain. In mice, ankyrin-G was required for forskolin-dependent reversal of depression-related behavior.
Conclusions
Taken together, ANK3 and DAGLA, both neuropsychiatric disorder genes, interact in a complex to regulate spine morphology. These data reveal novel synaptic signaling mechanisms and potential therapeutic avenues.







