Summary: Study could help pave the way for new research for neurodegenerative conditions such as motor neuron disease and muscular dystrophy.
Source: The Francis Crick Institute.
A study from scientists at the Francis Crick Institute, the Max-Delbrück Center for Molecular Medicine, Berlin and the University of Edinburgh sheds new light on the cells that form spinal cord, muscle and bone tissue in mammalian embryos.
This discovery paves the way for generating these tissues from stem cells in the laboratory and could lead to new ways of studying degenerative conditions such as motor neuron disease and muscular dystrophy.
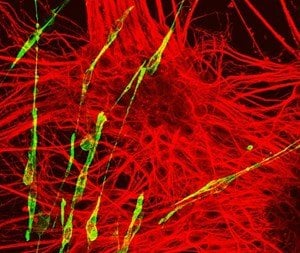
In embryos, the spinal cord, muscle and skeleton are produced from a group of cells called NMPs (neuro-mesodermal progenitors). These cells are few in number and exist only for a short time in embryos, despite giving rise to many tissues in the body. Their scarcity and inaccessibility has made studying NMPs challenging. Now, by using the latest molecular techniques, the research team has for the first time deciphered gene activity in NMPs. They used an advanced technique called single-cell transcriptional profiling, which analyses individual cells to provide a detailed picture of gene activity in every cell.
The technique allowed the team to establish a molecular signature of NMPs and to show that NMPs produced from stem cells in petri dishes in the laboratory closely resemble those found in embryos. This enabled the team to use lab-grown NMPs to learn more about these cells and how they make spinal cord, muscle and bone tissue. By manipulating the cells in petri dishes and testing the function of specific genes, the researchers re-constructed the regulatory mechanism and formulated a mathematical model that explains how NMPs produce the appropriate amounts of spinal cord and musculoskeletal cells.
Dr James Briscoe, who led the research from the Francis Crick Institute said:
“For embryonic development to progress smoothly, NMPs must make the right types of cells, in the right numbers at the right time. Understanding how cells such as NMPs make decisions is therefore central to understanding embryonic development. Single cell profiling techniques, including the ones we used in this study, are giving us unprecedented insight into this problem and offering a new and fascinating view of how embryos produce the different tissues that make up adults.”
First author of the study Dr Mina Gouti, from the Max-Delbrück Center for Molecular Medicine, Berlin said:
“Improving our understanding of NMPs doesn’t only answer an important developmental biology question but also holds great promise for regenerative medicine. It takes us a step closer to being able to use tissue from patients with diseases that affect muscles and motor neurons in order to study the causes and progress of these diseases. Being able to grow cells in the laboratory that faithfully resemble those found in the body is crucial for this.”
Source: The Francis Crick Institute
Image Source: NeuroscienceNews.com image is credited to James Briscoe, Francis Crick Institute.
Original Research: Full open access research for “A Gene Regulatory Network Balances Neural and Mesoderm Specification during Vertebrate Trunk Development” by Mina Gouti, Julien Delile, Despina Stamataki, Filip J. Wymeersch, Yali Huang, Jens Kleinjung, Valerie Wilson, and James Briscoe in Developmental Cell. Published online April 3 2017 doi:10.1016/j.devcel.2017.04.002
[cbtabs][cbtab title=”MLA”]The Francis Crick Institute “How Embryonic Cells Make Spinal Cord, Muscle and Bone.” NeuroscienceNews. NeuroscienceNews, 28 April 2017.
<https://neurosciencenews.com/embryonic-cells-spinal-cord-6541/>.[/cbtab][cbtab title=”APA”]The Francis Crick Institute (2017, April 28). How Embryonic Cells Make Spinal Cord, Muscle and Bone. NeuroscienceNew. Retrieved April 28, 2017 from https://neurosciencenews.com/embryonic-cells-spinal-cord-6541/[/cbtab][cbtab title=”Chicago”]The Francis Crick Institute “How Embryonic Cells Make Spinal Cord, Muscle and Bone.” https://neurosciencenews.com/embryonic-cells-spinal-cord-6541/ (accessed April 28, 2017).[/cbtab][/cbtabs]
Abstract
A Gene Regulatory Network Balances Neural and Mesoderm Specification during Vertebrate Trunk Development
Highlights
•Single-cell RNA-seq reveals a signature of neuromesodermal progenitors
•In vitro NMPs resemble and differentiate similar to their in vivo counterparts
•Dual role for retinoic acid signaling in NMP induction and neural differentiation
•A transcriptional network regulates neural versus mesodermal allocation
Summary
Transcriptional networks, regulated by extracellular signals, control cell fate decisions and determine the size and composition of developing tissues. One example is the network controlling bipotent neuromesodermal progenitors (NMPs) that fuel embryo elongation by generating spinal cord and trunk mesoderm tissue. Here, we use single-cell transcriptomics to identify the molecular signature of NMPs and reverse engineer the mechanism that regulates their differentiation. Together with genetic perturbations, this reveals a transcriptional network that integrates opposing retinoic acid (RA) and Wnt signals to determine the rate at which cells enter and exit the NMP state. RA, produced by newly generated mesodermal cells, provides feedback that initiates NMP generation and induces neural differentiation, thereby coordinating the production of neural and mesodermal tissue. Together, the data define a regulatory network architecture that balances the generation of different cell types from bipotential progenitors in order to facilitate orderly axis elongation.
“A Gene Regulatory Network Balances Neural and Mesoderm Specification during Vertebrate Trunk Development” by Mina Gouti, Julien Delile, Despina Stamataki, Filip J. Wymeersch, Yali Huang, Jens Kleinjung, Valerie Wilson, and James Briscoe in Developmental Cell. Published online April 3 2017 doi:10.1016/j.devcel.2017.04.002






