Inhibitory neuron functionality is not an immutable property of cortical cells, but a consequence of more complex network dynamics.
The two major types of neuron in the brain’s cerebral cortex are connected by intricate cortical circuits that process information. Excitatory neurons, which comprise 80 percent of all neurons in this region, increase activity in target cells. The other 20 percent of neurons are inhibitory, producing the opposite effect.
Inhibitory neurons (interneurons) have long been recognized as critical to understanding distinct kinds of information processing. Specific subtypes of interneurons can regulate response gain (the extent of reaction to stimuli) in excitatory neurons or shape response selectivity in target cells. In a new study scheduled appearing Dec. 11 in Nature Communications, Sami El-Boustani and Mriganka Sur of the Picower Institute for Learning and Memory hypothesize that a particular type of interneuron may influence responses in a context-dependent manner.
Arithmetic controls response
Inhibitory neurons in the visual cortex use division and subtraction to control computations performed by their target cells. Several previous studies suggested that one of the well-defined classes of interneuron, parvalbumin-expressing (PV +) cells, regulates response gain via divisive inhibition, whereas the other major class, somatostatin-positive (SOM +) cells, controls response selectivity via subtractive inhibition.
Divisive inhibition can occur during a range of functions including directed visual attention, orientation, multisensory integration, and value estimation. Subtractive inhibition is thought to sharpen neuronal selectivity, possibly increasing discrimination capability and therefore perhaps enhancing behavioral performance. Based on the findings of earlier research, these functions were thought to be native properties of these cell types, based on structure, connections, and biophysics.
Innovative techniques yield new findings
More recent studies, including an in-depth examination by El-Boustani and Sur, indicate that SOM + cells in particular are a key component of the underlying mechanisms that dictate functionality. In order to evoke different response dynamics in interneurons and investigate the nature of inhibition, the Picower neuroscientists employed “an innovative combination of novel visual stimuli, precisely-timed single-pulse optogenetic stimulation, and large-scale recording via two-photon calcium imaging of targeted neuronal responses in mice,” explains Sur, the Newton Professor of Neuroscience and director of the Simons Center for the Social Brain. Using briefly flashed visual stimuli and short pulses rather than prolonged optogenetic activation, the researchers were able to define the response times of PV + and SOM + neurons compared with their target cells, as well as isolate the inhibitory effect of these two types of neuron.
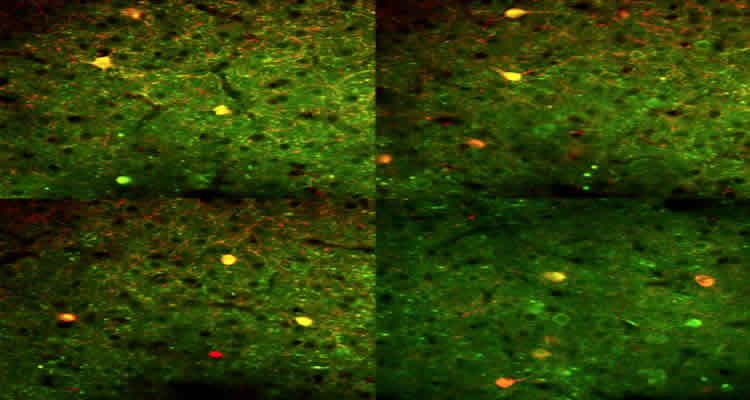
El-Boustani, a postdoctoral fellow in the Sur lab who led the study, was able to show that SOM + neurons can perform either divisive inhibition and regulate response gain or subtractive inhibition and control response selectivity. He found that when stimulation activates the SOM + cells at the same time as their target cells, as is the case when probed with large visual stimuli, they divide responses. When activation of the target cells occurs at a different time — sooner when they respond to small visual stimuli — the SOM + neurons subtract responses. “So neuronal functionality is dynamic; it is governed by firing coordination and the overlap of response timing in the circuits and their target cells,” explains El-Boustani.
The MIT scientists’ ability to demonstrate dramatic and distinct response-dependent switching in the live brain suggests that interneuron functionality is not an immutable property of each cell type, but a consequence of more complex dynamics within cortical networks. Since cortical inhibitory neurons mediate not only information processing but also play a critical role in brain disorders like autism, schizophrenia, and epilepsy, this discovery contributes significant insights to enhance understanding of normal and abnormal brain functionality.
Contact: Najat Kessler – MIT
Source: MIT press release
Image Source: The image is credited to Sami Elboustani and is adapted from the MIT press release
Original Research: Full open access research for “Response-dependent dynamics of cell-specific inhibition in cortical networks in vivo” by Sami El-Boustani and Mriganka Sur in Nature Communications. Published online December 11 2014 doi:10.1038/ncomms6689
Response-dependent dynamics of cell-specific inhibition in cortical networks in vivo
In the visual cortex, inhibitory neurons alter the computations performed by target cells via combination of two fundamental operations, division and subtraction. The origins of these operations have been variously ascribed to differences in neuron classes, synapse location or receptor conductances. Here, by utilizing specific visual stimuli and single optogenetic probe pulses, we show that the function of parvalbumin-expressing and somatostatin-expressing neurons in mice in vivo is governed by the overlap of response timing between these neurons and their targets. In particular, somatostatin-expressing neurons respond at longer latencies to small visual stimuli compared with their target neurons and provide subtractive inhibition. With large visual stimuli, however, they respond at short latencies coincident with their target cells and switch to provide divisive inhibition. These results indicate that inhibition mediated by these neurons is a dynamic property of cortical circuits rather than an immutable property of neuronal classes.
“Response-dependent dynamics of cell-specific inhibition in cortical networks in vivo” by Sami El-Boustani and Mriganka Sur in Nature Communications doi:10.1038/ncomms6689.