Summary: Brazilian scientists produce brain organoids with primitive eyes using computational fluid dynamics.
Source: D’Or Institute for Research and Education
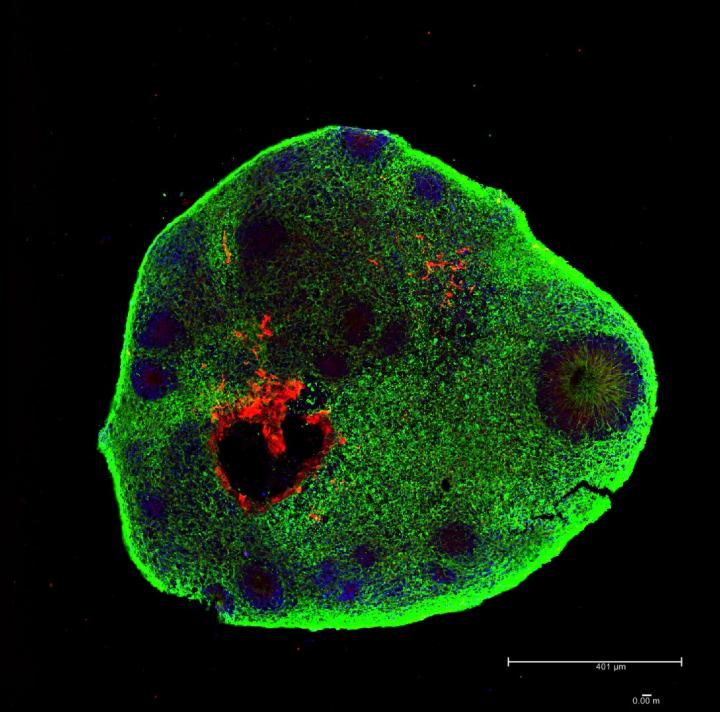
Scientists improved the initial steps of a standard protocol and produced organoids displaying regionalized brain structures, including retinal pigmented cells. The announcement was published in BMC Developmental Biology by the D’Or Institute for Research and Education’s team.
Human brain organoids are aggregates formed by nervous cells obtained from cell reprogramming. With this technique, cells extracted from skin or urine of volunteers are transformed into stem cells and then into neurons and other nervous cell types. They are cultivated for weeks, until they start forming agglomerates that resemble an embryonic brain.
For the past few years, scientists have been trying to perfect this model in order to create organoids increasingly complex and similar to those in later stages of development.
Since 2016, in partnership with the Federal University of Rio de Janeiro (UFRJ), scientists from the D’Or Institute have cultivated human brain organoids to study neurological diseases and the effects of new drugs on the nervous system.
They put nerve cells in a nutrient-rich liquid, similar to the development environment of the human embryo. From there, those mini-brains developed in a self-regulated process. In other words, all one must do is make sure they have the right environment to develop.
Recently, the team lead by Stevens Rehen has been able to refine the environment where the cells are maintained. “These organoids are a demonstration that it is possible to repeat, in the laboratory, increasingly advanced gradients of human brain development,” says Rehen. “We developed a cost-effective suspension method on orbital steering plates as an alternative for the cultivation of brain organoids with retinal pigmented cells”.
Source:
D’Or Institute for Research and Education
Media Contacts:
Daniel Rinaldi
Image Source:
D’Or Institute for Research and Education.
Original Research:
“Computational fluid dynamic analysis of physical forces playing a role in brain organoid cultures in two different multiplex platforms” in BMC Developmental Biology
10.1186/s12861-019-0183-y
Abstract
Computational fluid dynamic analysis of physical forces playing a role in brain organoid cultures in two different multiplex platforms
Background
Organoid cultivation in suspension culture requires agitation at low shear stress to allow for nutrient diffusion, which preserves tissue structure. Multiplex systems for organoid cultivation have been proposed, but whether they meet similar shear stress parameters as the regularly used spinner flask and its correlation with the successful generation of brain organoids has not been determined.
Results
Here we used computational fluid dynamics (CFD) to simulate two multiplex culture conditions: steering plates on an orbital shaker and the use of a previously described bioreactor. The bioreactor had low speed and high shear stress regions that may affect cell aggregate growth, depending on volume, whereas the computed variables of the steering plates were closer to those of the spinning flask.
Conclusion
Our protocol improves the initial steps of the standard brain organoid formation, and the produced organoids displayed regionalized brain structures, including retinal pigmented cells. Overall, we conclude that suspension culture on orbital steering plates is a cost-effective practical alternative to previously described platforms for the cultivation of brain organoids for research and multiplex testing.






