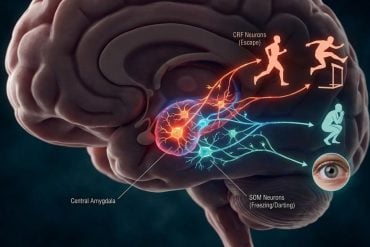
Summary: Relapse isn’t a failure of willpower—it’s a biological “rewiring” of the brain. A new study reveals how chronic cocaine use hijacks the connection between the brain’s reward center and its memory hub (the hippocampus).
Researchers identified a specific protein, DeltaFosB, that acts like a genetic master switch. As cocaine use continues, this protein accumulates, turning certain genes on and off to create a permanent, compulsive drive for the drug. By using CRISPR technology to pinpoint this mechanism, scientists have opened the door to a new class of “addiction-breaking” medications that could one day reset the brain’s circuitry and prevent relapse.
Key Facts
- The “Master Switch”: The protein DeltaFosB accumulates in the brain’s reward-memory circuit during chronic cocaine use, making it harder to quit and easier to relapse.
- Rewiring the Hippocampus: Cocaine use doesn’t just change how you feel; it physically alters the hippocampus, the brain’s memory center, linking drug-seeking behavior to deep-seated survival instincts.
- Calreticulin’s Role: Researchers found a secondary gene, calreticulin, that is controlled by DeltaFosB and helps “rev the brain’s engine” to seek out more cocaine.
- No Approved Meds: Currently, there are no FDA-approved medications to treat cocaine addiction. This research identifies DeltaFosB as a primary target for future pharmaceutical therapies.
- CRISPR Insights: Scientists used specialized CRISPR technology to prove that DeltaFosB isn’t just associated with addiction—it is necessary for the brain changes that drive it.
Source: Michigan State University
When a cocaine addict relapses, it isn’t a matter of personal failure — it’s the biological result of their brain’s rewiring, new research finds.
Michigan State University scientists found that cocaine changes how the hippocampus functions, contributing to the ongoing compulsion to seek out the drug. Their National Institutes of Health-supported research, published in Science Advances, not only explains why cocaine addiction is notoriously difficult to treat, but it could also help scientists develop new pharmaceutical therapies.
“Addiction is a disease in the same sense as cancer,” said senior author A.J. Robison, a professor of neuroscience and physiology. “We need to find better treatments and help people who are addicted in the same sense that we need to find cures for cancer.”
At least a million people nationwide struggle with cocaine addiction, and right now, there’s no FDA-approved medication to treat it. People who stop using don’t experience the same physical withdrawal symptoms that opiates cause, but that doesn’t mean it’s easy to quit. The drug hijacks the brain, flooding the reward centers with dopamine. This positive reinforcement tricks the brain into feeling like it’s doing something good instead of destructive.
Even if someone successfully quits, the odds aren’t in their favor. About 24% relapse to weekly use, and another 18% return to a treatment program within a year.
Andrew Eagle, a former postdoctoral researcher in Robison’s lab and the paper’s lead author, found a key player responsible for the compulsion — a protein called DeltaFosB. He used a specialized form of CRISPR technology to examine the role this protein plays in specific brain circuits when mice were exposed to cocaine.
Using mouse models, he learned that this protein acts like a switch, turning genes on and off in the circuit between the brain’s reward center and the hippocampus, the brain’s memory hub. The longer someone uses cocaine, the more this protein accumulates in the circuit. This protein changes how the neurons function, altering how the circuit responds to cocaine.
“This protein isn’t just associated with these changes, it is necessary for them,” Eagle said. “Without it, cocaine does not produce the same changes in brain activity or the same strong drive to seek out the drug.”
The research team also found another group of genes controlled by DeltaFosB after chronic cocaine use. One of those genes, called calreticulin, helps regulate how neurons communicate with each other. Their work showed calreticulin contributes to revving the brain’s engine to compulsively seek out more cocaine.
These findings in mouse models could have direct applications to humans, which share many of the same genes and similar circuits. Robison’s lab is partnering with researchers at the University of Texas Medical Branch in Galveston, Texas, to create compounds that target DeltaFosB. Together, they have a grant from the National Institute of Drug Abuse to develop and test compounds that regulate DeltaFosB’s ability to bind to DNA.
“If we could find the right kind of compound that works in the right way, that could potentially be a treatment for cocaine addiction,” Robison said. “That’s years away, but that’s the long-term goal.”
Next, Robison’s lab will examine how hormones impact these brain circuits, and whether cocaine affects the male and female brain differently. This work could help explain biological differences in addiction risk between men and women.
Key Questions Answered:
A: This research confirms it is a biological disease. Chronic use physically alters the brain’s genetic expression, creating a protein buildup that forces the brain to prioritize the drug over everything else. It’s a “rewiring” that willpower alone often cannot overcome.
A: Because the drug hijacks your memory center (the hippocampus). The protein DeltaFosB creates a long-lasting “switch” in your neural circuits that stays flipped long after the drug has left your system, keeping the drive to seek it out active.
A: While we aren’t there yet, researchers are currently developing compounds specifically designed to block DeltaFosB from binding to DNA. If successful, these could “reset” the addicted brain and make long-term recovery much more achievable.
Editorial Notes:
- This article was edited by a Neuroscience News editor.
- Journal paper reviewed in full.
- Additional context added by our staff.
About this addiction and neuroscience research news
Author: Bethany Mauger
Source: Michigan State University
Contact: Bethany Mauger – Michigan State University
Image: The image is credited to Neuroscience News
Original Research: Open access.
“Transcriptional regulation of ventral hippocampus-nucleus accumbens circuit excitability drives cocaine seeking” by Andrew L. Eagle, Chiho Sugimoto, Marie A. Doyle, Daniela Anderson, Seyedeh Leila Mousavi, Megan M. Dykstra, Hayley M. Kuhn, Brooklynn R. Murray, Ryan M. Bastle, Sarah Simmons, Jin He, Ian Maze, Michelle S. Mazei-Robison, and Alfred J. Robison. Science Advances
DOI:10.1126/sciadv.adv1236
Abstract
Transcriptional regulation of ventral hippocampus-nucleus accumbens circuit excitability drives cocaine seeking
Ventral hippocampus (vHPC) CA1 pyramidal neurons send glutamatergic projections to nucleus accumbens (NAc), and this vHPC-NAc circuit mediates cocaine seeking and reward, but it is unclear whether vHPC-NAc neuron properties are modulated by cocaine exposure to drive subsequent behavior.
The immediate early gene transcription factor FosB/ΔFosB is induced throughout the brain by cocaine and is critical for cocaine seeking, but its function in vHPC-NAc neurons is not understood.
We now show that circuit-specific knockout of FosB/ΔFosB in vHPC-NAc neurons impaired cocaine reward expression and forced abstinence–induced seeking. We also found that vHPC-NAc excitability was decreased by experimenter-administered repeated cocaine and cocaine self-administration, and this cocaine-induced excitability decrease was mediated by ΔFosB expression.
To uncover the mechanism of this change in circuit function, we used circuit-specific translating ribosome affinity purification to assess cocaine-induced, FosB/ΔFosB-dependent changes in gene expression in vHPC-NAc. We found that cocaine causes a FosB/ΔFosB-dependent increase in the expression of calreticulin, an endoplasmic reticulum–resident calcium-buffering protein.
Calreticulin expression mediated vHPC-NAc excitability and was necessary for cocaine reward. These findings uncover a noncanonical mechanism by which cocaine increases calreticulin in vHPC leading to decreased vHPC-NAc excitability and drives cocaine seeking and reward.