Summary: Too many chandelier cells in the binocular region of the brain impair visual depth perception.
Source: Cold Spring Harbor Laboratory
Our perceptions of the world are shaped both by our biology and our experiences. New work from Cold Spring Harbor Laboratory Adjunct Professor Z. Josh Huang provides a glimpse of how these factors intersect in a part of the brain that integrates visual information, establishing the neural circuitry that literally shapes how we see the world.
The study, published in the journal Neuron, concerns the connections that integrate information from the left and right eyes to generate a single, coherent image. The initial wiring of the brain sets the stage for this information processing early in development, but visual experiences are important for fine-tuning the circuits. “We know it’s not whether it’s nature or nurture, but it’s clearly both,” Huang says. “The question is really how does nature constrain nurture, or how does the nurture work with nature?”
In their study, Huang and colleagues focused on so-called chandelier cells, elaborately branched neurons that reach out to dampen the signaling of hundreds of neighboring cells. They resemble an old-fashioned chandelier with many branches. These cells, which are some of the most potent inhibitory cells in the nervous system, are distributed with some regularity throughout the brain. But in the part of the brain that generates binocular vision, their density thins.
Genetically engineered mice developed in the Huang lab allowed the team to visualize these cells in the brain. Strikingly, they found that the cells were much more abundant within the binocular region of the visual cortex immediately after birth than they were in the brains of older animals. The process of pruning chandelier cells starts in the retina at the back of the eye. The young retina sends signals to the visual cortex that kill off the inhibitory cells. Huang explains:
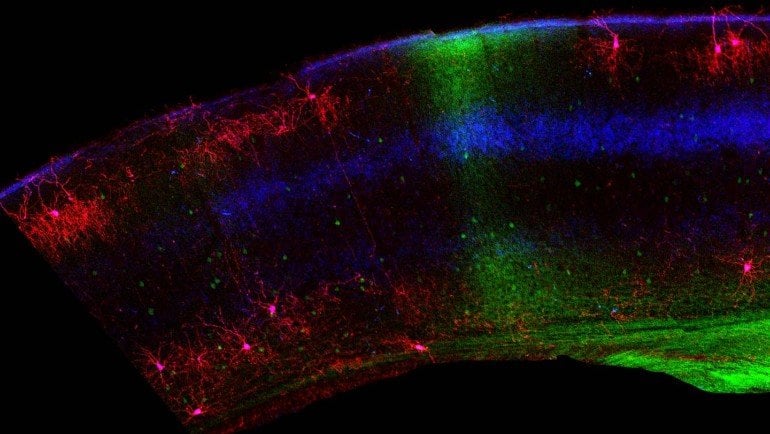
“In just a matter of a few days, before the mouse opens their eyes, so before their experience, there’s a very dramatic process that eliminates these cells. More than half of them are basically killed.”
Reducing chandelier cells’ numbers likely frees cells in the binocular region of the visual cortex to communicate more efficiently between the two visual hemispheres, which is essential for rapidly integrating signals relayed from both eyes, Huang says. In fact, when the team interfered with this process, generating mice where chandelier cells remained abundant in this part of the brain, the animals’ depth perception was severely impaired, suggesting an inability to integrate the information taken in by the two eyes.
About this visual neuroscience news
Source: Cold Spring Harbor Laboratory
Contact: Sara Roncero-Menendez – Cold Spring Harbor Laboratory
Image: The image is credited to Bor-Shuen Wang, Huang lab
Original Research: Closed access.
“Retinal and Callosal Activity-Dependent Chandelier Cell Elimination Shapes Binocularity in Primary Visual Cortex” by Huang et al. Neuron
Abstract
Retinal and Callosal Activity-Dependent Chandelier Cell Elimination Shapes Binocularity in Primary Visual Cortex
Highlights
- •Massive chandelier cell (ChC) apoptosis in the binocular zone prior to vision onset
- •Callosal input from contralateral visual cortex drives ChC elimination
- •Spontaneous retinal activities prior to vision regulate ChC elimination
- •Preventing ChCs elimination results in deficient binocularity and visual behavior
Summary
In mammals with binocular vision, integration of the left and right visual scene relies on information in the center visual field, which are relayed from each retina in parallel and merge in the primary visual cortex (V1) through the convergence of ipsi- and contralateral geniculocortical inputs as well as transcallosal projections between two visual cortices. The developmental assembly of this binocular circuit, especially the transcallosal pathway, remains incompletely understood. Using genetic methods in mice, we found that several days before eye-opening, retinal and callosal activities drive massive apoptosis of GABAergic chandelier cells (ChCs) in the binocular region of V1. Blockade of ChC elimination resulted in a contralateral eye-dominated V1 and deficient binocular vision. As pre-vision retinal activities convey the left-right organization of the visual field, their regulation of ChC density through the transcallosal pathway may prime a nascent binocular territory for subsequent experience-driven tuning during the post-vision critical period.






