Summary: Non-invasive auricular transcutaneous vagus nerve stimulation (atVNS), applied to an accessible area of the ear, improves memory in mouse models of intellectual disability.
Source: UPF Barcelona
Researchers at UPF are testing a new non-invasive method of stimulating the vagus nerve in mice that improves their memory. They have shown for the first time that electrostimulation in the ear of intellectual disability rodent models leads to a cognitive improvement.
The study is the result of a collaboration between research groups at the Department of Experimental and Health Sciences (DCEXS) and the Department of Information and Communication Technologies (DTIC). It has been coordinated by the researcher Andrés Ozaita, principal investigator at the Neuropharmacology Laboratory-NeuroPhar, and is published in the journal Brain Stimulation.
Stimulation of the vagus nerve emerged as a therapy to treat epilepsy and depression that do not respond to drugs, because the ramifications of this nerve carry information to deregulated areas of the brain in both diseases. Some of these regions like the amygdala, prefrontal cortex, and hippocampus, are also required for attention and memory. For this reason, the technique has also been found to produce cognitive improvement.
Initially, stimulation was carried out invasively, surgically implanting the device that applies electrical impulses. Subsequently, non-invasive approaches were developed, which stimulate the skin’s surface in areas in which the branches of the nerve pass.
Previous studies had revealed the modulation of memory using invasive and non-invasive approaches in animal models and in humans, but non-invasive transcutaneous (through the skin) approaches had not been tested so far in mouse models.
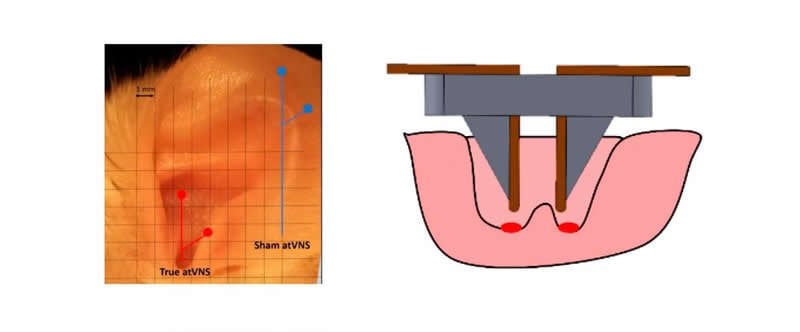
Researchers at the Biomedical Electronics Research Group developed an electrode to allow non-invasive electrostimulation of the vagus nerve in mice. “Mikel Domingo-Gainza, a student of the bachelor’s degree in Biomedical Engineering, built the electrode during his bachelor’s degree final project”, explains Antoni Ivorra, leader of the Biomedical Electronics Research Group (BERG) and DTIC lecturer. The device is applied in an accessible area of the mouse’s ear, which is reached by branches of the vagus nerve.
“We show that this technology achieves a behavioral effect of cognitive enhancement in mice”, explains Anna Vázquez-Oliver, co-first author of the article. “We use a test to evaluate the memory in which we value whether the mouse remembers objects it is familiar with. After electrostimulation, the animals achieved better results in the test, their memory lasted longer”, she adds. This would be the first proof in mice; in rats it had been shown that applying electrostimulation to the ear yielded brain responses –more neurotransmitter activity–, but no effect on behavior had been demonstrated.
They then tested the potential of the protocol in a mouse model of fragile X syndrome, the most common form of inherited intellectual disability, which usually shows poor performance in object recognition memory. Transcutaneous stimulation also improved their memory, which supports its relevance in cognitive modulation in the intellectual disability mice model.
In the words of Cecilia Brambilla-Pisoni, co-first author of the study, “the activity we are generating artificially in the fibers of the vagus nerve would produce an activation of areas of the brain that are important for memory. We hypothesize that an increased release of noradrenaline would occur, making the information better consolidated”.
Andrés Ozaita notes that “the results confirm the potential of this therapeutic tool that is worth exploring in the context of neurodevelopmental disorders, as previously proposed for invasive forms”. Knowing that this technique is functional opens up an important field at the pre-clinical level, since it can be applied to animal models of other diseases. “Now we are focusing on the cellular and molecular results produced by this intervention to be able to elucidate the mechanisms involved”, he concludes.
Funding: Anna Vázquez-Oliver has a predoctoral grant from the Training Programme of the Jérôme Lejeune Foundation to investigate in the treatment of intellectual disability. Cecilia Brambilla-Pisoni received an Inphinit scholarship from La Caixa Foundation. The study has been funded by a Marie Slodowska-Curie scholarship, from the Ministry of Economy, Innovation and Competitiveness (MINECO), the Carlos III Health Institute, the Catalan Government, ICREA, the “María de Maeztu Unit of Excellence”, and the European Regional Development Fund (ERDF).
Source:
UPF Barcelona
Media Contacts:
UPF – UPF Barcelona
Image Source:
The image is credited to UPF.
Original Research: Open access
“Auricular transcutaneous vagus nerve stimulation improves memory persistence in naïve mice and in an intellectual disability mouse model”. Vázquez-Oliver A, Brambilla-Pisoni C, Domingo-Gainza M, Maldonado R, Ivorra A, Ozaita A.
Brain Stimulation doi:10.1016/j.brs.2019.12.024.
Abstract
Auricular transcutaneous vagus nerve stimulation improves memory persistence in naïve mice and in an intellectual disability mouse model
Highlights
• Auricular transcutaneous vagus nerve stimulation was performed in anesthetized mice.
• atVNS enhanced non-emotional memory persistence in naïve mice.
• atVNS improved memory performance in the Fmr1 KO mouse model for fragile X syndrome.
Abstract
Background
Vagus nerve stimulation (VNS) using non-invasive approaches have attracted great attention due to their anti-epileptic, anti-depressive and pro-cognitive effects. It has been proposed that auricular transcutaneous VNS (atVNS) could benefit intellectual disability disorders, but preclinical data supporting this idea is limited.
Objective
To develop an atVNS device for mice and to test its efficacy on memory performance in naïve mice and in a mouse model for intellectual disability.
Methods
Naïve outbreed CD-1 mice and a model for fragile X syndrome, the Fmr1 knockout (Fmr1KO), were used to assess the effect of atVNS in the novel object-recognition memory performance.
Results
We found that atVNS significantly improves memory persistence in naïve mice. Notably, atVNS was efficacious in normalizing the object-recognition memory deficit in the Fmr1KO model.
Conclusion
Our data show that atVNS improves memory persistence in naïve mice and in a model of intellectual disability and support further studies taking advantage of preclinical mouse models of cognitive disorders.






