Summary: New findings reveal phagocytes do not fully mature until after birth, contradicting previous assumptions that they mature during embryonic development.
Source: University of Freiberg
What gets into the brain and what doesn’t is strictly regulated. Researchers at the Faculty of Medicine at the University of Freiburg have now studied phagocytes that coat the blood vessels in the brain and reinforce the blood-brain barrier.
As the scientists from the Institute of Neuropathology at the Medical Center—University of Freiburg together with an international research team have shown, these cells only mature fully after birth according to a defined step-by-step developmental program.
Until now, it had been assumed that this process was completed during embryonic development.
Their studies, which were published in the journal Nature on April 20, 2022, were initially carried out on genetically modified mouse lines and were confirmed on human samples. They are expected to provide important insights into the development and treatment of diseases of the brain.
“We were able to show that the immune cells we studied migrate from the cerebral membrane to the blood vessels in the brain shortly before birth and mature there. This process is probably not completed until weeks after birth and could partly explain why the brain is so vulnerable at the beginning of life,” says Prof. Dr. Marco Prinz, Medical Director of the Institute of Neuropathology at the Medical Center—University of Freiburg and head of the Collaborative Research Center/Transregio 167—NeuroMac and member of the Cluster of Excellence CIBSS -Centre for Integrative Biological Signalling Studies at the University of Freiburg.
“The late timing of the maturation of the phagocytes, also called macrophages, was very surprising to us, since the precursor cells are already present in the brain long before,” says Prinz.
In addition, the scientists were able to show for the first time that the vessels, as structure-giving cells of the brain, send important signals for normal development of the brain’s macrophages.
The blood-brain barrier is formed by cells on the blood vessels of the brain. They control which substances can enter the brain and which cannot. This protects the brain from harmful substances and pathogens. The blood-brain barrier is particularly permeable in the case of infectious diseases, certain brain tumors and oxygen deficiency.
Significance for Alzheimer’s, multiple sclerosis and more
“In addition to the blood-brain barrier, the immune cells we studied control what can reach the brain cells from the blood, they eat pathogens and prevent excessive inflammation. They are also involved in the development of cancer, Alzheimer’s disease and multiple sclerosis. Our findings could be important for a better understanding of these diseases and future therapies,” Prinz adds.
Color-coded cells and gene analyses
For their study, the researchers led by the two first authors Dr. Takahiro Masuda from Kyushu University, Japan, and Dr. Lukas Amann from the Faculty of Medicine at the University of Freiburg used several newly established mouse lines.
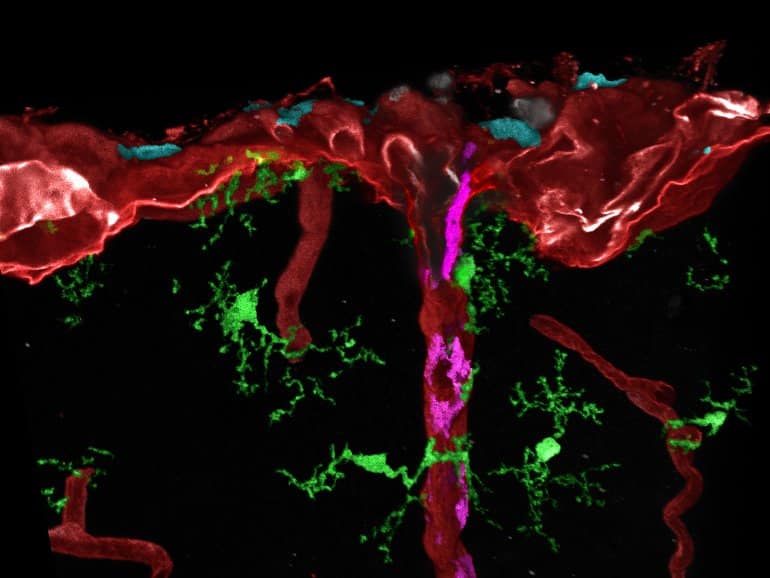
With these, different types of brain macrophages and their progenitor cells could be specifically labeled for the first time and later found in the different brain regions using high-resolution microscopy. In addition, they examined the gene activity of individual cells and thus determined their degree of maturity.
“We were also able to confirm the data on human brains. This gives us a much deeper understanding of the timing and molecular mechanisms in the development of the cells. This knowledge can now be used to explore new and more specific therapeutic approaches for brain diseases,” says biologist Dr. Lukas Amann, who works at the Institute of Neuropathology at the Medica Center—University of Freiburg.
About this neuroscience research news
Author: Press Office
Source: University of Freiberg
Contact: Press Office – University of Freiberg
Image: The image is credited to University of Freiburg / Dr. Lukas Amann
Original Research: Closed access.
“Specification of CNS macrophage subsets occurs postnatally in defined niches” by Lukas Amann et al. Nature
Abstract
Specification of CNS macrophage subsets occurs postnatally in defined niches
All tissue-resident macrophages of the central nervous system (CNS)—including parenchymal microglia, as well as CNS-associated macrophages (CAMs) such as meningeal and perivascular macrophages—are part of the CNS endogenous innate immune system that acts as the first line of defence during infections or trauma.
It has been suggested that microglia and all subsets of CAMs are derived from prenatal cellular sources in the yolk sac that were defined as early erythromyeloid progenitors.
However, the precise ontogenetic relationships, the underlying transcriptional programs and the molecular signals that drive the development of distinct CAM subsets in situ are poorly understood.
Here we show, using fate-mapping systems, single-cell profiling and cell-specific mutants, that only meningeal macrophages and microglia share a common prenatal progenitor.
By contrast, perivascular macrophages originate from perinatal meningeal macrophages only after birth in an integrin-dependent manner. The establishment of perivascular macrophages critically requires the presence of arterial vascular smooth muscle cells.
Together, our data reveal a precisely timed process in distinct anatomical niches for the establishment of macrophage subsets in the CNS.






