Summary: Altering the structure of perineuronal nets could be a mechanism that underlies sleep-induced memory changes.
Source: SfN
The morphing structure of the brain’s “cartilage cells” may regulate how memories change while you snooze, according to new research in eNeuro.
Sleep lets the body rest, but not the brain. During sleep, the brain accounts for a day of learning by making strong memories stronger and weak memories weaker, a process known as memory consolidation. But changing memories requires changing synapses, the connections between neurons. Sleep-induced changes need to overcome perineuronal nets, cartilage-like sheaths that not only surround and protect neurons, but also prevent changes in synapses.
Pantazopoulos et al. investigated how perineuronal nets varied during sleep in mice. By documenting whether or not they could tag the nets with a protein that binds to a specific sugar chain, they could observe the changes in synapses. A decrease in the number of tagged nets would indicate an increase in the number of neurons allowing synaptic changes.
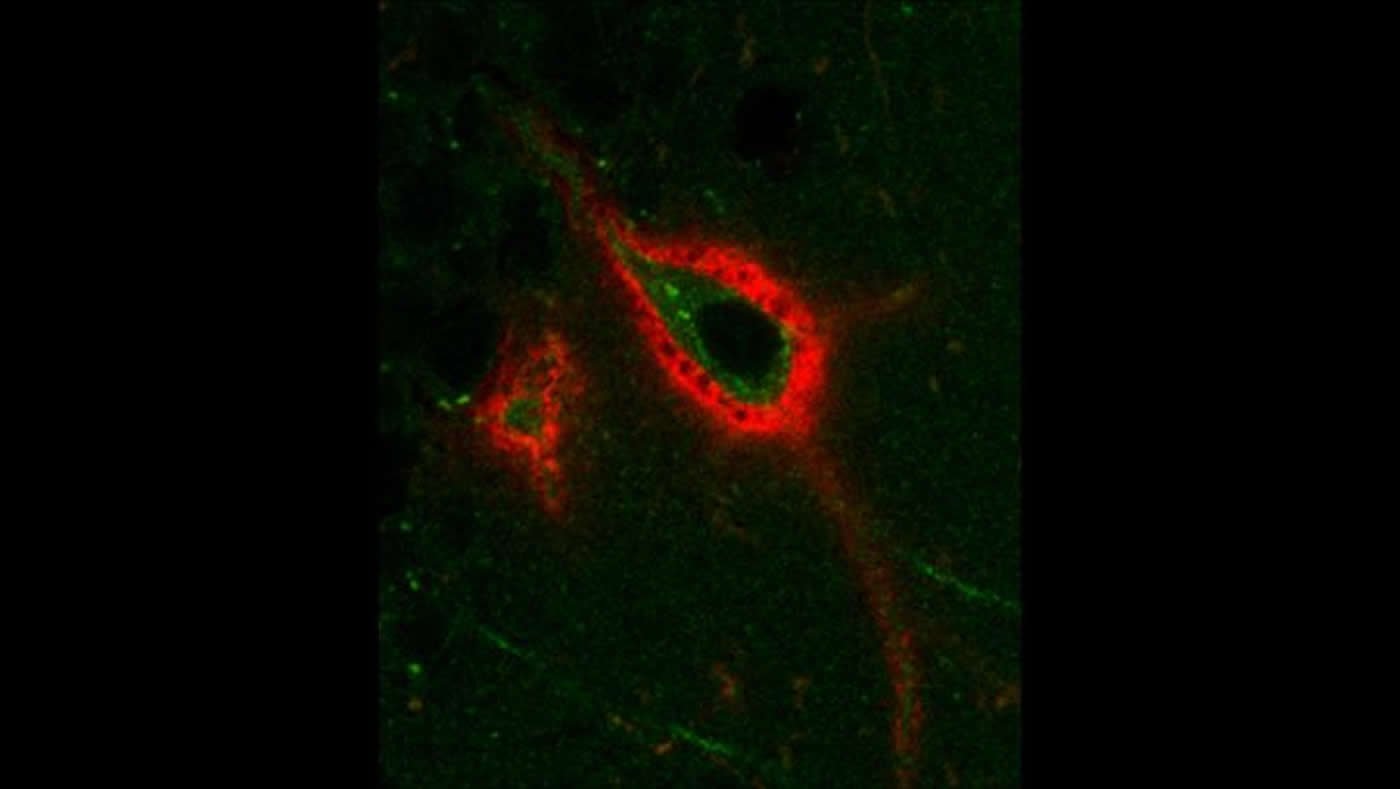
Tagging increased during wakefulness and decreased during sleep. Sleep deprivation prevented this change. The levels of a net-altering enzyme expressed by brain immune cells cycled in opposition, hinting that it may be responsible for the change. The research team also compared levels of tagged nets in human brain tissue with the donor’s time of death. Human brains displayed similar sleep-centric rhythms in net structure. Altering the structure of perineuronal nets may be one of the mechanisms behind sleep-induced memory changes.
About this learning and sleep research article
Source:
SfN
Media Contacts:
Calli McMurray – SfN
Image Source:
The image is credited to Pantazopoulos et al., eNeuro 2020.
Original Research: Closed access
“Circadian Rhythms of Perineuronal Net Composition”. by Harry Pantazopoulos, Barbara Gisabella, Lindsay Rexrode, David Benefield, Emrah Yildiz, Phoebe Seltzer, Jake Valeri, Gabriele Chelini, Anna Reich, Magdalena Ardelt and Sabina Berretta. eNeuro.
Abstract
Circadian Rhythms of Perineuronal Net Composition
Perineuronal Nets (PNNs) are extracellular matrix (ECM) structures that envelop neurons and regulate synaptic functions. Long thought to be stable structures, PNNs have been recently shown to respond dynamically during learning, potentially regulating the formation of new synapses. We postulated that PNNs vary during sleep, a period of active synaptic modification. Notably, PNN components are cleaved by matrix proteases such as the protease cathepsin-S. This protease is diurnally expressed in the mouse cortex, coinciding with dendritic spine density rhythms. Thus, cathepsin-S may contribute to PNN remodeling during sleep, mediating synaptic reorganization. These studies were designed to test the hypothesis that PNN numbers vary in a diurnal manner in the rodent and human brain, as well as in a circadian manner in the rodent brain, and that these rhythms are disrupted by sleep deprivation. In mice, we observed diurnal and circadian rhythms of PNNs labeled with the lectin wisteria floribunda agglutinin (WFA+PNNs) in several brain regions involved in emotional memory processing. Sleep deprivation prevented the daytime decrease of WFA+ PNNs and enhances fear memory extinction. Diurnal rhythms of cathepsin-S expression in microglia were observed in the same brain regions, opposite to PNN rhythms. Finally, incubation of mouse sections with cathepsin-S eliminated PNN labeling. In humans, WFA+PNNs showed a diurnal rhythm in the amygdala and thalamic reticular nucleus (TRN). Our results demonstrate that PNNs vary in a circadian manner and this is disrupted by sleep deprivation. We suggest that rhythmic modification of PNNs may contribute to memory consolidation during sleep.
Significance Statement
The mechanisms underlying memory consolidation are not completely understood. Perineuronal nets are extracellular matrix structures enveloping subsets of neurons and are involved in regulating synaptic plasticity. Recent studies indicate that perineuronal nets are modified during learning to allow for formation of new synapses. During sleep, synapses are proposed to undergo modification as memory consolidation processes occur. Furthermore, microglia are involved in synaptic regulation and produce several proteases that cleave perineuronal net components. We demonstrate that perineuronal nets are modified in a circadian manner and coincide with expression rhythms of the protease cathepsin-S. These rhythms may contribute to altered synaptic plasticity reported during sleep, suggesting a key process through which proteases modify PNNs to allow for to memory consolidation.