Summary: Researchers report transplanting bone marrow from young mice to older mice helps to preserve memory and learning skills. If the findings can be replicated in humans, researchers believe new therapies can be devised to help combat disorders associated with brain aging.
Source: Cedars Sinai Medical Center.
A new study has found that transplanting the bone marrow of young laboratory mice into old mice prevented cognitive decline in the old mice, preserving their memory and learning abilities. The findings support an emerging model that attributes cognitive decline, in part, to aging of blood cells, which are produced in bone marrow.
“While prior studies have shown that introducing blood from young mice can reverse cognitive decline in old mice, it is not well understood how this happens,” said Helen Goodridge, PhD, associate professor of Medicine and Biomedical Sciences at Cedars-Sinai and co-senior author of the study. “Our research suggests one answer lies in specific properties of youthful blood cells.”
If further research confirms similar processes in people, the findings could provide a pathway for designing therapies to slow progression of neurodegenerative diseases, including Alzheimer’s, that affect millions of Americans, Goodridge said.
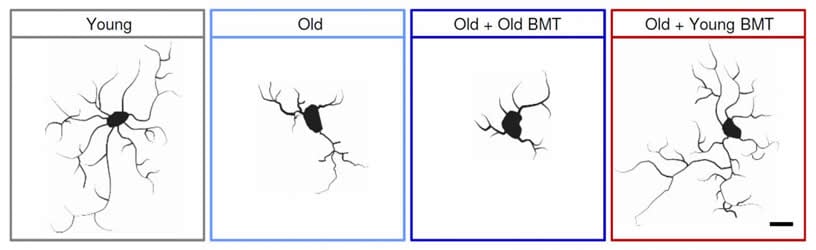
In the study, published in the journal Communications Biology, 18-month-old laboratory mice received bone marrow transplants from either 4-month-old mice or mice their own age. Six months later, both transplanted groups underwent standard laboratory tests of activity level and learning, plus spatial and working memory. Mice that received young bone marrow outperformed mice that received old bone marrow. They also outperformed a control group of old mice that did not get transplants.
The research team then examined the hippocampus, a region associated with memory, in the mice brains. Recipients of young bone marrow retained more connections, known as synapses, between neurons in the hippocampus than did recipients of old bone marrow, even though they had about the same number of neurons. Synapses are critical to brain performance.
Further tests showed a possible reason for the missing synapses. The blood cells made by the young bone marrow reduced the activation of microglia, a type of immune cell in the brain. Microglia support neuron health but can become overactive and participate in disconnection of the synapses. With fewer overactive microglia, neurons would remain healthy and more synapses would survive.
“We are entering an era in which there will be more elderly people in the population, along with an increased incidence of Alzheimer’s disease, putting a huge burden on the health system,” said Clive Svendsen, PhD, director of the Cedars-Sinai Board of Governors Regenerative Medicine Institute, professor of Biomedical Sciences and Medicine and co-senior author of the new study. “Our work indicates that cognitive decline in mice can be significantly reduced by simply providing young blood cells, which act on the brain to reduce the loss of synapses related to aging.”
Translating the findings, if confirmed in human samples, into potential treatments may be challenging, given that bone marrow transplants are not currently feasible for this use. But for future studies in people, Svendsen is working on creating “personalized” young blood stem cells for an individual through stem cell technology. These cells possibly could be used to help replace the individual’s own aging blood stem cells and help prevent cognitive decline and perhaps neurodegenerative diseases such as Alzheimer’s as well.
Source: Jane Engle – Cedars Sinai Medical Center
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Cedars-Sinai / Communications Biology.
Original Research: Open access research for “Young bone marrow transplantation preserves learning and memory in old mice” by Melanie M. Das, Marlesa Godoy, Shuang Chen, V. Alexandra Moser, Pablo Avalos, Kristina M. Roxas, Ivy Dang, Alberto Yáñez, Wenxuan Zhang, Catherine Bresee, Moshe Arditi, George Y. Liu, Clive N. Svendsen & Helen S. Goodridge in Communications Biology. Published February 20 2019.
doi:10.1038/s42003-019-0298-5
[cbtabs][cbtab title=”MLA”]Cedars Sinai Medical Center”Young Bone Marrow Rejuvenates Aging Brains: Mouse Study.” NeuroscienceNews. NeuroscienceNews, 20 February 2019.
<https://neurosciencenews.com/bone-marrow-brain-aging-10778/>.[/cbtab][cbtab title=”APA”]Cedars Sinai Medical Center(2019, February 20). Young Bone Marrow Rejuvenates Aging Brains: Mouse Study. NeuroscienceNews. Retrieved February 20, 2019 from https://neurosciencenews.com/bone-marrow-brain-aging-10778/[/cbtab][cbtab title=”Chicago”]Cedars Sinai Medical Center”Young Bone Marrow Rejuvenates Aging Brains: Mouse Study.” https://neurosciencenews.com/bone-marrow-brain-aging-10778/ (accessed February 20, 2019).[/cbtab][/cbtabs]
Abstract
Young bone marrow transplantation preserves learning and memory in old mice
Restoration of cognitive function in old mice by transfer of blood or plasma from young mice has been attributed to reduced C–C motif chemokine ligand 11 (CCL11) and β2-microglobulin, which are thought to suppress neurogenesis in the aging brain. However, the specific role of the hematopoietic system in this rejuvenation has not been defined and the importance of neurogenesis in old mice is unclear. Here we report that transplantation of young bone marrow to rejuvenate the hematopoietic system preserved cognitive function in old recipient mice, despite irradiation-induced suppression of neurogenesis, and without reducing β2-microglobulin. Instead, young bone marrow transplantation preserved synaptic connections and reduced microglial activation in the hippocampus. Circulating CCL11 levels were lower in young bone marrow recipients, and CCL11 administration in young mice had the opposite effect, reducing synapses and increasing microglial activation. In conclusion, young blood or bone marrow may represent a future therapeutic strategy for neurodegenerative disease.






