Researchers from Trinity College Dublin have discovered that specialised ‘astrocytes’ abandon their posts as protectors and repairers of the brain when primed by the neurodegeneration of cells around them.
Astrocytes, named as a result of their star-shaped appearance, function in healthy brains to assist with normal brain function and metabolism.
However, the researchers from Trinity’s School of Biochemistry and Immunology have now demonstrated that these cells play a major role in the inflammatory hypersensitivity of the degenerating brain, by responding in an inappropriate and exaggerated fashion when stimulated by inflammation around them.
This discovery has important implications for the management of neurodegenerative conditions such as stroke and Parkinson’s and Alzheimer’s diseases.
In findings just published in the Journal of Neuroscience, the research group of Wellcome Trust Research Professor, Colm Cunningham, showed that stimulated astrocytes produce exaggerated amounts of molecules known as chemokines, which mobilise and recruit large numbers of inflammatory cells from the periphery to the brain. Normally, this process is tightly regulated in the young, healthy brain.
The chemokines CCL2 and CXCL1 are synthesised when the brain is exposed to acute pro-inflammatory cytokines, such as those produced during acute injury to the brain or during neurodegeneration.
These same chemokines were produced at much greater levels, however, when astrocytes of the neurodegenerating brain were similarly challenged. This in turn led to a large infiltration of neutrophils and monocytes, which act as key parts of the immune system, but which can contribute to negative brain outcomes when infiltration is uncontrolled and inflammation is excessive.
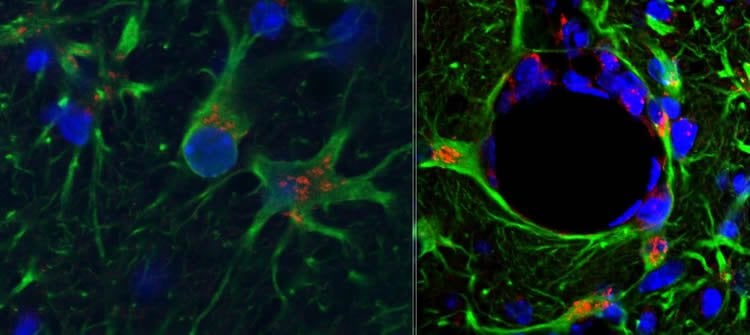
Crucially, this discovery expands our knowledge of the inflammatory vulnerability of the aging and degenerating brain and hints at strategies that could lessen this susceptibility.
Professor Cunningham said: “If one recognises the mechanisms by which the brain becomes susceptible to the effects of co-morbidities and environmental insults, one can then lessen their effects, irrespective of whether these strategies target the primary pathology. It is possible that targeting chemokines, or the inflammatory cells that produce them, may have protective effects in vulnerable patients.”
Funding: Professor Cunningham is funded by a Wellcome Trust Senior Research Fellowship, while Edel Hennessy, a PhD student who performed much of the research, was supported by a Trinity Foundation College Award.
Source: Thomas Deane – Trinity College Dublin
Image Credit: The image is credited to the researchers
Original Research: Full open access research for “Astrocytes Are Primed by Chronic Neurodegeneration to Produce Exaggerated Chemokine and Cell Infiltration Responses to Acute Stimulation with the Cytokines IL-1β and TNF-α” by Edel Hennessy, Éadaoin W. Griffin, and Colm Cunningham in Journal of Neuroscience. Published online June 3 2015 doi:10.1523/JNEUROSCI.2745-14.2015
Abstract
Astrocytes Are Primed by Chronic Neurodegeneration to Produce Exaggerated Chemokine and Cell Infiltration Responses to Acute Stimulation with the Cytokines IL-1β and TNF-α
Microgliosis and astrogliosis are standard pathological features of neurodegenerative disease. Microglia are primed by chronic neurodegeneration such that toll-like receptor agonists, such as LPS, drive exaggerated cytokine responses on this background. However, sterile inflammatory insults are more common than direct CNS infection in the degenerating brain and these insults drive robust IL-1β and TNF-α responses. It is unclear whether these pro-inflammatory cytokines can directly induce exaggerated responses in the degenerating brain. We hypothesized that glial cells in the hippocampus of animals with chronic neurodegenerative disease (ME7 prion disease) would display exaggerated responses to central cytokine challenges. TNF-α or IL-1β were administered intrahippocampally to ME7-inoculated mice and normal brain homogenate-injected (NBH) controls. Both IL-1β and TNF-α produced much more robust IL-1β synthesis in ME7 than in NBH animals and this occurred exclusively in microglia. However, there was strong nuclear localization of the NFκB subunit p65 in the astrocyte population, associated with marked astrocytic synthesis of the chemokines CXCL1 and CCL2 in response to both cytokine challenges in ME7 animals. Conversely, very limited expression of these chemokines was apparent in NBH animals similarly challenged. Thus, astrocytes are primed in the degenerating brain to produce exaggerated chemokine responses to acute stimulation with pro-inflammatory cytokines. Furthermore, this results in markedly increased neutrophil, T-cell, and monocyte infiltration in the diseased brain. These data have significant implications for acute sterile inflammatory insults such as stroke and traumatic brain injury occurring on a background of aging or neurodegeneration.






