Summary: Study provides evidence of the existence of a long-rage inhibitory pathway from the auditory cortex to the amygdala in the brains of mice.
Source: UT San Antonio
Researchers at The University of Texas at San Antonio (UTSA) are pioneering an innovative brain study that sheds light on how the amygdala portion of the brain functions and could contribute to a better understanding of post-traumatic stress disorder, anxiety, depression and Alzheimer’s disease.
The researchers, Alfonso Apicella, an associate professor in the Department of Biology, research associate Alice Bertero, postdoctoral fellow Paul Luc Caroline Feyen, and graduate student Hector Zurita, published their study, “A non-canonical cortico-amygdala inhibitory loop,” in the Journal of Neuroscience. Their research was also featured on the Oct. 23 cover of the journal.
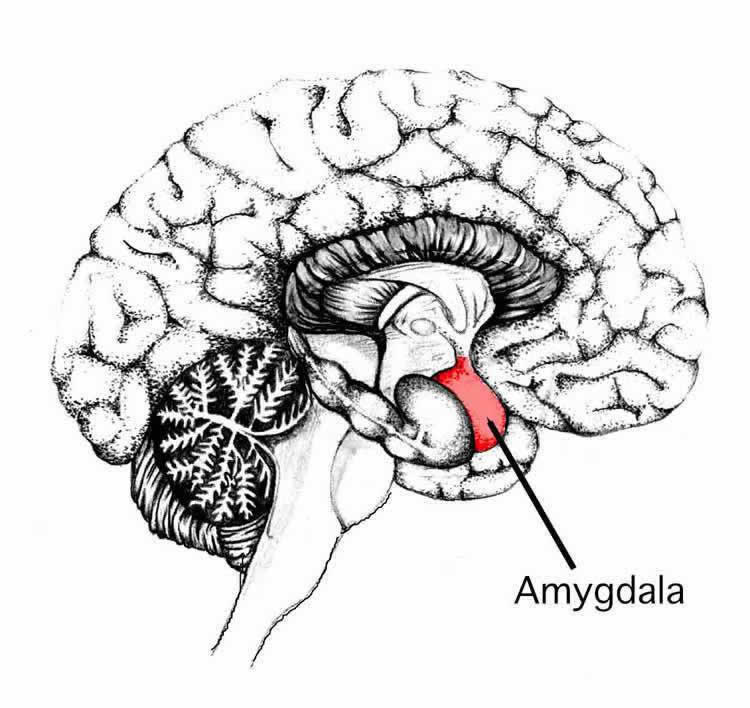
Conditions such as PTSD, anxiety and depression are thought to be linked to the abnormal functioning of the amygdala, which is located within the temporal lobes and plays a key role in processing emotions, actions and cognition.
“This novel research paper provides anatomical and physiological evidence for the existence of a long-range inhibitory pathway from the auditory cortex to the amygdala in the mouse brain,” Apicella said. “For the first time, in our paper we show this emotional pathway.”
Apicella explained that advances in the techniques for labeling individual neuronal cells made it possible to study the individual neurons extending from the auditory cortex to the amygdala.
He added that the inhibitory cortical neurons can alter the activity of the amygdala’s principal neurons and can therefore directly control the output of the amygdala. The neurons contain a substance called somatostatin, and it regulates physiological functions and forms a connection with principal neurons that project to other brain regions outside the amygdala that are involved in fear and aversive behavior.
“The discovery that the amygdala receives both excitatory and inhibitory inputs from that cortex suggests that the timing and relative strength of these inputs can affect the activity of the amygdala,” Acipella said.
Apicella and his research team noted that future experiments should examine whether this is a general mechanism by which sensory stimuli can influence the processes controlled by the amygdala, such as fear/aversive behavior and how the disruption of this pathway can lead to several neurological and psychiatric disorders, such as,Alzheimer’s, anxiety, depression and PTSD.
Research related to this topic will continue in Apicella’s lab. His research group investigates the neural basis of perception. More specifically, the researchers want to understand how cortical microcircuits process sensory information leading to behavioral outcomes.
Source:
UT San Antonio
Media Contacts:
Kara Soria – UT San Antonio
Image Source:
The image is in the public domain.
Original Research: Closed access
“A Non-Canonical Cortico-Amygdala Inhibitory Loop”. Alice Bertero, Paul Luc Caroline Feyen, Hector Zurita and Alfonso junior Apicella.
Journal of Neuroscience doi:10.1523/JNEUROSCI.1515-19.2019.
Abstract
A Non-Canonical Cortico-Amygdala Inhibitory Loop
Discriminating between auditory signals of different affective value is critical for the survival and success of social interaction of an individual. Anatomical, electrophysiological, imaging, and optogenetics approaches have established that the auditory cortex (AC) by providing auditory information to the lateral amygdala (LA) via long-range excitatory glutamatergic projections has an impact on sound-driven aversive/fear behavior. Here we test the hypothesis that the LA also receives GABAergic projections from the cortex. We addressed this fundamental question by taking advantage of optogenetics, anatomical, and electrophysiology approaches and directly examining the functional effects of cortical GABAergic inputs to LA neurons of the mouse (male/female) AC. We found that the cortex, via cortico-lateral-amygdala somatostatin neurons (CLA-SOM), has a direct inhibitory influence on the output of the LA principal neurons. Our results define a CLA long-range inhibitory circuit (CLA-SOM inhibitory projections → LA principal neurons) underlying the control of spike timing/generation in LA and LA–AC projecting neurons, and attributes a specific function to a genetically defined type of cortical long-range GABAergic neurons in CLA communication.
SIGNIFICANCE STATEMENT
It is very well established that cortical auditory inputs to the lateral amygdala are exclusively excitatory and that cortico-amygdala neuronal activity has been shown to be involved in sound-driven aversive/fear behavior. Here, for the first time, we show that the lateral amygdala receives long-range GABAergic projection from the auditory cortex and these form direct monosynaptic inhibitory connections onto lateral amygdala principal neurons. Our results define a cellular basis for direct inhibitory communication from auditory cortex to the lateral amygdala, suggesting that the timing and ratio of excitation and inhibition, two opposing forces in the mammalian cerebral cortex, can dynamically affect the output of the lateral amygdala, providing a general mechanism for fear/aversive behavior driven by auditory stimuli.






