Summary: Researchers have developed a new rat model of Alzheimer’s disease that can be used to study the accumulation of amyloid plaques.
Source: University of Missouri Columbia.
Alzheimer’s disease currently affects more than 5.5 million Americans and is one of the costliest diseases to treat, according to the Alzheimer’s Association. Characterized by a buildup of plaque in the brain, few animal models exist that researchers could use to study this devastating disorder. Now, a team of researchers from the University of Missouri, publishing in PLOS ONE, developed a rat model that can be used to study the buildup of amyloid plaques and vascular abnormalities in the brain.
“One of the defining traits of Alzheimer’s is the progressive accumulation of amyloid-β plaques in the brain,” said Yuksel Agca, associate professor of veterinary pathology and a researcher in the Comparative Medicine Program in the MU College of Veterinary Medicine. “Two proteins, APP and PS1 are linked to the development of Alzheimer’s disease in humans–and these two targets have become the basis for numerous studies. If we can identify how to manipulate amyloid-β build up, we can reduce the production of harmful plaques in the brain, leading to decreases in the onset of Alzheimer’s disease.”
The MU researchers developed rats that had human APP and PS1 proteins. The rats then underwent behavioral assessments in the Barnes maze. The maze, which tracks rodent behavior is used to measure learning and spatial memory. In a series of experiments, the Alzheimer’s model rats that produced human APP and PS1 displayed poor memory and learning.
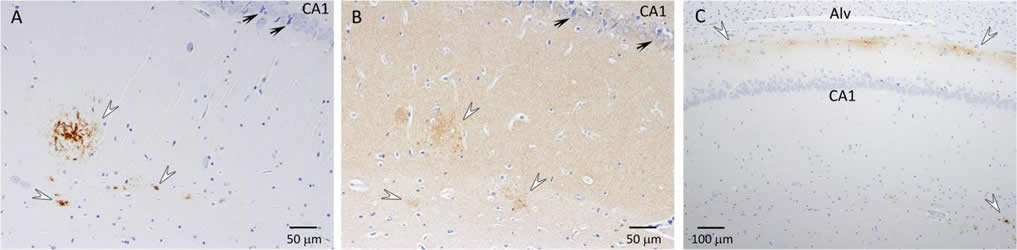
After the maze experiments, the research team assessed amyloid-β levels through serum tests, as well as brain screenings, which showed vascular changes and amyloid-β plaques similar to the ones observed in humans suffering from Alzheimer’s disease.
“We found that these rat models could be useful in the fight against Alzheimer’s in people,” Agca said. “Because of their shorter lifespans and their larger size, translational models such as rats are extremely helpful in ongoing studies of disease. The results can be translatable to humans in identifying targets for drugs as well as identifying everyday lifestyle changes we can make to help stave off disorders like Alzheimer’s. We hope this model will become beneficial as research continues to move forward.”
Just like plaque on teeth or in arteries, plaque buildup in the brain can be decreased in people by maintaining a good diet and exercise, Agca said.
The study, “Memory deficiency, cerebral amyloid angiopathy, and amyloid-β plaques in APP+PS1 double transgenic rat model of Alzheimer’s disease,” was published in PLOS ONE. This study was supported by the University of Missouri-Research Incentive Funds to Yuksel Agca and the National Institutes of Health (# P40 OD011062). Diana Klakotskaia, a graduate student in the Department of Psychological Science in the MU College of Arts and Science; and Cansu Agca, a researcher in the Department of Veterinary Pathobiology contributed to the study. The content is solely the responsibility of the authors and does not necessarily represent the official views of the funding agencies.
Funding: Funding for this study was provided by the National Institute of Health.
Source: Jeff Sossamon – University of Missouri Columbia
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Yuksel Agca, MU College of Veterinary Medicine.
Original Research: Open access research for “Memory deficiency, cerebral amyloid angiopathy, and amyloid-β plaques in APP+PS1 double transgenic rat model of Alzheimer’s disease” by Diana Klakotskaia, Cansu Agca, Rachel A. Richardson, Edward G. Stopa, Todd R. Schachtman, and Yuksel Agca in PLOS ONE. Published April 11 2018.
doi:10.1371/journal.pone.0195469
[cbtabs][cbtab title=”MLA”]University of Missouri Columbia “New Model Could Uncover Important Answers in Alzheimer’s Research.” NeuroscienceNews. NeuroscienceNews, 30 April 2018.
<https://neurosciencenews.com/alzheimers-model-8925/>.[/cbtab][cbtab title=”APA”]University of Missouri Columbia (2018, April 30). New Model Could Uncover Important Answers in Alzheimer’s Research. NeuroscienceNews. Retrieved April 30, 2018 from https://neurosciencenews.com/alzheimers-model-8925/[/cbtab][cbtab title=”Chicago”]University of Missouri Columbia “New Model Could Uncover Important Answers in Alzheimer’s Research.” https://neurosciencenews.com/alzheimers-model-8925/ (accessed April 30, 2018).[/cbtab][/cbtabs]
Abstract
Memory deficiency, cerebral amyloid angiopathy, and amyloid-β plaques in APP+PS1 double transgenic rat model of Alzheimer’s disease
Transgenic rat models of Alzheimer’s disease were used to examine differences in memory and brain histology. Double transgenic female rats (APP+PS1) over-expressing human amyloid precursor protein (APP) and presenilin 1 (PS1) and single transgenic rats (APP21) over-expressing human APP were compared with wild type Fischer rats (WT). The Barnes maze assessed learning and memory and showed that both APP21 and APP+PS1 rats made significantly more errors than the WT rats during the acquisition phase, signifying slower learning. Additionally, the APP+PS1 rats made significantly more errors following a retention interval, indicating impaired memory compared to both the APP21 and WT rats. Immunohistochemistry using an antibody against amyloid-β (Aβ) showed extensive and mostly diffuse Aβ plaques in the hippocampus and dense plaques that contained tau in the cortex of the brains of the APP+PS1 rats. Furthermore, the APP+PS1 rats also showed vascular changes, including cerebral amyloid angiopathy with extensive Aβ deposits in cortical and leptomeningeal blood vessel walls and venous collagenosis. In addition to the Aβ accumulation observed in arterial, venous, and capillary walls, APP+PS1 rats also displayed enlarged blood vessels and perivascular space. Overall, the brain histopathology and behavioral assessment showed that the APP+PS1 rats demonstrated behavioral characteristics and vascular changes similar to those commonly observed in patients with Alzheimer’s disease.






